Academic Courses
FACULTY OF CLINICAL MEDICINE AND SURGERY
Click here to access Unit one Content..
Topic 1.1: What is an STI?
The term "sexually transmitted diseases" is used to denote disorders spread principally by intimate sexual contact.
There are other diseases that can be spread via sexual contact and other means e.g brucellosis, ebola etc. These diseases are however not classified as STI as sexual contact is not their primary mode of transmission.
Although sex usually means sexual intercourse, it also includes;
- Close body contact,
- Kissing,
- Cunnilingus,
- Anilingus,
- Fellatio,
- Mouth–breast contact, and
- Anal intercourse.
Many sexually transmitted diseases (STDs) can also be acquired by;Transplacental spread, during;
- Passage through the birth canal, and
- Via lactation during the neonatal period.
Role of Clinicians in STI management;
- To understand the microbiology of STDs in order to appropriately diagnose and treat patients.
- To alleviate the symptoms and prevent future sequelae.
- To prevent the transmission to others including health care professionals.
- Patient education and counseling.
Prevention through lifestyle and behavioral modification is the primary weapon against the spread of STDs.
Multiple cohort studies have demonstrated the protective effects of both male and female condoms.
Click here to access Unit one Content..
Topic 1.1: What is an STI? [Cont'd]
Common causes of STIs.
The list of organisms traditionally thought of as causing STDs has been extended to include;
- Cytomegalovirus,
- Herpes simplex virus type 1 (HSV-1) and type 2 (HSV-2),
- Chlamydia,
- Group B Streptococcus,
- Molluscum contagiosum virus,
- Sarcoptes scabiei,
- Hepatitis viruses, and
- Human immunodeficiency virus (HIV).
- Some diseases spread by body contact but not necessarily by coitus—eg, pediculosis pubis and molluscum contagiosum.
Risk factors for STI;
- UTI especially by STI pathogens.
- Female gender.
- Multiple unprotected sexual contacts.
- Sex worker.
- Intravenous drug users.
- New relationship in the last 2 months.
General Signs and symptoms of STI;
Ay patient who present with the following symptoms should be investigated for possible STI.
- Sores or bumps on the genitals or in the oral or rectal area
- Painful or burning urination
- Discharge from the penis
- Unusual or odd-smelling vaginal discharge
- Unusual vaginal bleeding
- Pain during sex
- Sore, swollen lymph nodes, particularly in the groin but sometimes more widespread
- Lower abdominal pain in females.
- Fever
- Rash over the trunk, hands or feet.
Specimens for investigating STIs;
- Blood; Whole blood. Serum.
- Urine.
- Vaginal swab; High vaginal swab, Low vaginal swab.
- Rectal swab.
- Throat swab.
- Urethral/vaginal secretions.
- Histological specimen
Click here to access Unit one Content..
Topic 1.2: Classification and General features of STIs.
CLASSIFICATION OF STDS;
1. Aetiological Classification;
1) Bacterial
i. Chancroid -Haemophilus ducreyi.
ii. Chlamydia -Chlamydia trachomatis
iii. Gonorrhea -Neisseria gonorrhoeae.
iv. Granuloma inguinale or -Klebsiella granulomatis.
v. Syphilis -Treponema pallidum
2) Fungal
i. Candidiasis (yeast infection)
3) Viral.
i. Viral hepatitis (Hepatitis B virus)—saliva, venereal fluids.
(Note: Hepatitis A and Hepatitis E are transmitted via the fecal-oral route; Hepatitis C is rarely sexually transmittable, and the route of transmission of Hepatitis D (only if infected with B) is uncertain, but may include sexual transmission.
ii. Herpes simplex (Herpes simplex virus 1, 2) skin and mucosal, transmissible with or without visible blisters
iii. HIV (Human Immunodeficiency Virus)—venereal fluids, semen, breast milk, blood
iv. HPV (Human Papillomavirus)—skin and mucosal contact. 'High risk' types of HPV cause almost all cervical cancers, as well as some anal, penile, and vulvar cancer. Some other types of HPV cause genital warts.
v. Molluscum contagiosum (molluscum contagiosum virus MCV)—close contact
4) Parasites.
i. Crab louse, colloquially known as "crabs" or "pubic lice" (Pthirus pubis). The infestation and accompanying inflammation is Pediculosis pubis
ii. Scabies (Sarcoptes scabiei)
iii. Trichomoniasis (Trichomonas vaginalis), colloquially known as "trich"
2. Clinical classification.
1) Ulcer forming/Genital Ulcer Disease (GUD).
i. Herpes simples.
ii. Syphilis/Chancre.
iii. Chancroid.
iv. Condylomata acuminate/venereal warts.
v. Granuloma Inguinale/Donovanosis.
vi. Lymphogranuloma venereum LGV.
General features of GUD (Genital Ulcer Disease);
An ulcer on the genitalia;
- Glans penis or penile shaft.
- Labia majora or minora.
- Vaginal ulcer.
The ulcer is painful in haemophilus ducreyi, herpes genitalis.
Painless in syphyllis.
2) Non Ulcer Forming.
i. Genital warts.
ii. Condylomata acuminate.
VULVAR LESIONS & GENITAL ULCERS;
1. Genital herpes,
2. Syphilis, and
3. Chancroid are the most prevalent ulcerative lesions.
The diagnosis is difficult to make by physical examination alone.
The work-up for all genital ulcers should include serologic screening for
1) Syphilis,
2) Culture/antigen testing for HSV-1 and HSV-2, and
3) Culture for Haemophilus ducreyi.
More than one infectious etiology may be present in a single lesion. In today's environment it is important to recognize HIV as a risk factor for genital ulcers.
Click here to access Unit one Content..
Topic 1.3: Chancroid (Soft chancre)
Chancroid is an STD characterized by a painful genital ulcer.
However, studies have shown asymptomatic carriers among commercial sex workers.
Although this condition can be difficult to diagnosis clinically, suppurative inguinal adenopathy with painful ulcers is pathognomonic and may assist with a preculture diagnosis.

Aetiology;
Gram-negative rod H ducreyi.
Exposure is usually through coitus, but accidentally acquired lesions of the hands have occurred.
The incubation period is short: the lesion usually appears in 3–5 days or sooner. An increased rate of HIV infection has been reported among patients with this genital ulcer disease; chancroid is a cofactor for HIV transmission.
Moreover, 10% of patients with genital chancroid may have coinfection with herpes or syphilis.
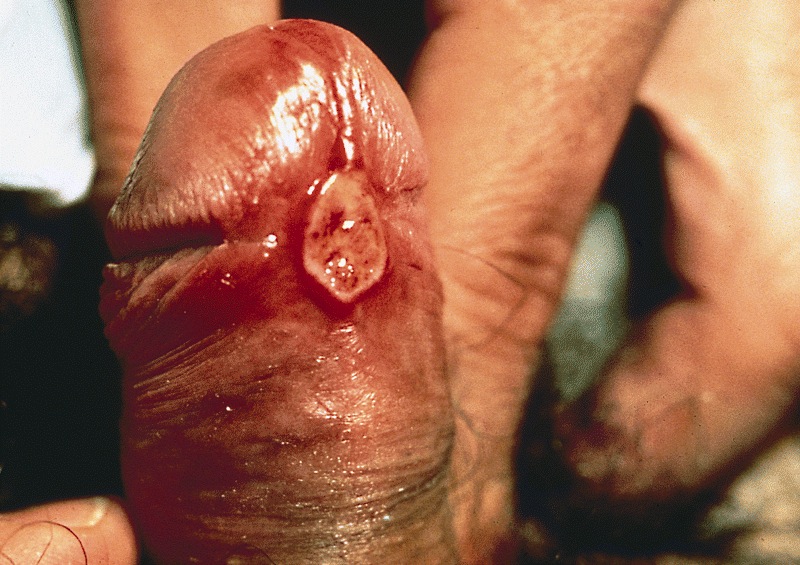
Clinical Features of Chancroid;
1. Vesico pustule on the pudendum, vagina, or cervix. Later, it degenerates into a saucer-shaped ragged ulcer circumscribed by an inflammatory wheal.
2. Typically, the lesion is very tender and produces a heavy, foul discharge that is contagious.
3. A cluster of ulcers may develop. Lesions typically occur on the vulva, cervix, and perianal area in women.
4. Painful inguinal adenitis is noted in over 50% of cases. The buboes may become necrotic and drain spontaneously.

Laboratory Dx;
Syphilis must first be ruled out.
1) Demonstration of the organism in pathological specimens (Vulval smear- from the ulcer, pus)
i. Clinical diagnosis is more reliable than smears or cultures because of the difficulty of isolating this organism.
ii. Isolation of H ducreyi is diagnostic, but isolation occurs in less than one-third of cases.
iii. Aspirated pus from a bubo is the best material for culture.
iv. Serum adsorption enzyme immunoassays have been evaluated and currently have a limited sensitivity.
v. Polymerase chain reaction (PCR) testing of genital samples is becoming widely available.
vi. Multiplex PCR is a technique that can simultaneously screen for HSV, Treponemapallidum, and H ducreyi using a single swab but is not yet commercially available.
2) RPR-Rapid Plasminogen Reagen to rule out syphilis.

Differential Diagnosis of chancroid;
1. Syphilis,
2. Granuloma inguinale,
3. Lymphogranuloma venereum,
4. Herpes simplex may coexist with chancroid and must be ruled out.
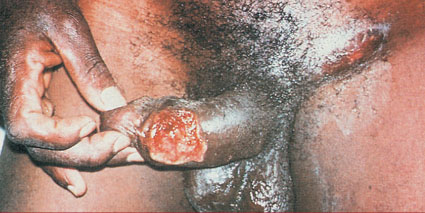
Prevention of chancroid;
Chancroid is a notifiable disease in some settings.
Routine antibiotic prophylaxis is not warranted.
Condoms can give protection.
Liberal use of soap and water is relatively effective. Education is essential.
4Cs
i. Counselling and education.
ii. Condom promotion.
iii. Compliance to medication.
iv. Contact tracing.
Treatment of chancroid;
1) Good personal hygiene is important.
a. The early lesions should be cleansed with mild soap solution.
b. Sitz baths are beneficial.
2) Antibiotic Treatment
Guidelines issued by the Centers for Disease Control and Prevention (CDC) for genital chancroid are as follows.
Recommended regimen;
i. Azithromycin 1 g orally once;
ii. Ceftriaxone 250 mg intramuscularly (IM) as a single dose;
iii. Erythromycin 500 mg orally 3 times daily for 7 days;
iv. Ciprofloxacin 500 mg orally twice daily for 3 days in nonpregnant patients over age 17 years who are not lactating. The course may have to be repeated.
v. Fluctuant lymph nodes may need to be aspirated through normal adjacent skin. Incision and drainage of the nodes is not recommended because it will delay healing.
Prognosis
Untreated or poorly managed cases of chancroid may persist, and secondary infection may develop.
Frequently, the ulcers heal spontaneously. They should improve within 7–10 days. If no improvement is noted, coinfection, HIV, resistant strains, and noncompliance must be considered. If not treated, they may cause deep scarring with sequelae in men.
Click here to access Unit one Content..
Topic 1.4: Granuloma Inguinale (Donovanosis)
Granuloma inguinale is a chronic ulcerative granulomatous disease that usually develops in the vulva, perineum, and inguinal regions.
Aetiology;
The causative organism is Calymmatobacterium granulomatis (Donovan body).
Donovan bodies are bacteria encapsulated in mononuclear leukocytes.
Transmission is via coitus, and the incubation period is 8–12 weeks.
Clinical features of Granuloma inguinale;
Although granuloma inguinale most often involves the skin and subcutaneous tissues of the vulva and inguinal regions, cervical, uterine, orolabial, and ovarian sites have been reported.
1. A malodorous discharge is characteristic.
2. The disorder often begins as a papule, which then ulcerates, with the development of a beefy-red granular zone with clean, sharp edges. The ulcer shows little tendency to heal, and the patient usually has no local or systemic symptoms. Healing is very slow, and satellite ulcers may unite to form a large lesion.
3. Lymphatic permeation is rare, but lymphadenitis may result when the cutaneous lesion becomes superimposed on lymphatic channels. Inguinal swelling is common, with late formation of abscesses (buboes).
4. Rarely, granuloma inguinale is manifested by chronic cervical lesions. These lesions usually take the form of redness or ulceration, or they form granulation tissue.
5. They produce a chronic inflammatory exudate characterized histologically by lymphocytes, giant cells, and histiocytes. They may mimic carcinoma of the cervix and must be distinguished from this as well as other neoplastic diseases.
6. The chronic ulcerative process may involve the urethra and the anal area, causing marked discomfort.
7. Introital contraction may make coitus difficult or impossible; walking or sitting may become painful. The possibility of the coexistence of another venereal disease must be considered. Spread to other areas occurs in approximately 7% of patients.
Laboratory Findings;
1. Direct smear from beneath the surface of an ulcer may reveal gram-negative bipolar rods within mononuclear leukocytes. These are seen best in Wright-stained smears.
2. When smears are negative, a biopsy specimen should be taken. Biopsy of the lesion generally shows granulation tissue infiltrated by plasma cells and scattered large macrophages with rod-shaped cytoplasmic inclusion bodies (Mikulicz cells).
3. Pseudoepitheliomatous hyperplasia often is seen at the margin of the ulcer.
4. Demonstrating, in biopsy or smear material stained with Wright's, Giemsa's, or silver stain, large mononuclear cells having one or more cystic inclusions containing the so-called Donovan bodies—small round or rod-shaped particles that stain purple in traditional hematoxylin and eosin preparations.
Prevention of Granuloma inguinale;
Personal hygiene is the best method of prevention. Therapy immediately after exposure may abort the infection.
Rx of granuloma inguinale;
1. Trimethoprim-sulfamethoxazole (CTX) 960mg BD X 3/52
2. Doxycycline 100 mg BD X 3/52.
3. Ciprofloxacin 750 mg BD X 3/52.
4. Erythromycin 500 QID X 2–3/52.
5. Azithromycin 1 g OD per week for 3 weeks.
Penicillin is not effective.
Sex partners must be considered for treatment.
Partners who had sexual contact during the 60 days preceding the onset of symptoms or are clinically symptomatic should be treated by 1 of the regimens.
Special consideration for HIV and gravid women should be made. Recommendations to add intravenous gentamicin to the oral protocol have been made.
Click here to access Unit one Content..
Topic 1.5: Lymphogranuloma Venereum (LGV)
Aetiology;
The causative agent of lymphogranuloma venereum is 1 of the aggressive L serotypes (L1, L2, or L3) of Chlamydia trachomatis.
Transmission is via sexual contact; men are affected more frequently than are women (6:1). The incubation period is 7–21 days.
Clinical features;
1. A vesicopustular eruption which may go undetected.
2. Inguinal (and vulvar) ulceration,
3. Lymphedema,
4. Genital pain.
5. During the inguinal bubo phase, the groin is exquisitely tender.
6. A hard cutaneous induration (red to purplish-blue) is a notable feature. This usually occurs within 10–30 days after exposure and may be bilateral.
7. Anorectal lymphedema occurs early; defecation is painful, and the stool may be blood-streaked.
8. Vaginal narrowing and distortion may end in severe dyspareunia.
9. In the late phase, systemic symptoms;
i. Fever,
ii. Headache,
iii. Arthralgia,
iv. Chills,
v. Abdominal cramps.
Laboratory Features of LGV.
1. Isolating C trachomatis from appropriate specimens and confirming the immunotype. These procedures are seldom available, so less specific tests are used.
2. A complement fixation test using a heat-stable antigen that is group-specific for all Chlamydia species is available. This test is positive at a titer 1:16 in more than 80% of cases of lymphogranuloma venereum. If acute or convalescent sera are available, a rise in titer is particularly helpful in making the diagnosis. Application of the microimmunofluorescent test may also be useful.
Differential Diagnosis of LGV;
As with any disseminated disease, the systemic symptoms of lymphogranuloma venereum may resemble;
1. Meningitis,
2. Arthritis,
3. Pleurisy,
4. Peritonitis.
5. Granuloma inguinale,
6. Tuberculosis,
7. Early syphilis,
8. Chancroid.
In the case of colonic lesions, proctoscopic examination and mucosal biopsy are needed to rule out carcinoma, schistosomiasis, and granuloma inguinale.
Complications of LGV;
1) Perianal scarring
2) Rectal strictures—late complications—can involve the entire sigmoid, but the urogenital diaphragm is rarely involved.
3) Vulvar elephantiasis (esthiomene) produces marked distortion of the external genitalia.
Prevention;
Avoiding infectious contact with a carrier is achieved by use of a condom or by refraining from coitus.
Definite exposure can be treated with sulfonamides or tetracyclines.
Rx of LGV;
1. Chemotherapy
1) Doxycycline 100 mg bd x 21/7.If disease persists, the course should be repeated.
2) Erythromycin 500 mg QID X 21/7.
2. Local and Surgical Treatment
1) Anal strictures should be dilated manually at weekly intervals.
2) Severe stricture may require diversionary colostomy.
3) If the disease is arrested, complete vulvectomy may be done for cosmetic reasons.
4) Abscesses should be aspirated, not excised.
Click here to access Unit one Content..
Topic 1.6: Syphyllis
Aetiology;
Treponema pallidum;
Forms of Syphyllis;
1) Primary Syphilis
i. Painless genital sore (chancre) on labia, vulva, vagina, cervix, anus, lips, or nipples.
ii. Painless, rubbery, regional lymphadenopathy followed by generalized lymphadenopathy in the third to sixth weeks.
Laboratory findings in primary syphyllis;
· Dark-field microscopic findings.
· Positive serologic test in 70% of cases.
2) Secondary Syphilis
i. Bilaterally symmetric extragenital papulosquamous eruption.
ii. Condyloma latum, mucous patches.
iii. Lymphadenopathy.
Laboratory findings in secondary syphyllis;
· Dark-field findings positive in moist lesions.
· Positive serologic test for syphilis.
3) Tertiary Syphilis
i. Cardiac,
ii. Neurologic,
iii. Ophthalmic, and
iv. Auditory lesions.
v. Gummas.
4. Congenital Syphilis
1) History of maternal syphilis.
2) Positive serologic test for syphilis.
3) Stigmata of congenital syphilis (eg, x-ray changes of bone, hepatosplenomegaly, jaundice, anemia).
4) Normal examination or signs of intrauterine infection.
5) Often stillborn or premature.
6) Enlarged, waxy placenta.
The course of syphilis is unaltered by pregnancy, but misdiagnoses are common.
Syphylitic chancre in congenital syphilis;
The chancre is often unnoticed or internal and not brought to medical attention. Chancres, mucous patches, and condyloma lata are often thought to be herpes genitalis. The dermatoses can resolve prior to diagnosis, or they may be misdiagnosed.
The effect of syphilis on pregnancy outcome can be profound.
Risk of congenital infection;
The risk of fetal infection depends on;
1. The degree of maternal spirochetemia (greater in the secondary stage than in the primary or latent stages).
2. The gestational age of the fetus.
Treponemes may cross the placenta at all stages of pregnancy, but fetal involvement is rare before 18 weeks because of fetal immunoincompetence.
After 18 weeks, the fetus is able to mount an immunologic response, and tissue damage may result. The earlier in pregnancy the fetus is exposed, the more severe the fetal infection and the greater the risk of premature delivery or stillbirth.
Antepartum infection in late pregnancy does not necessarily result in congenital infection, as only 40–50% of such infants will have definite congenital infection. Placental infection can occur with resultant endarteritis, stromal hyperplasia, and immature villi.
Placenta in congenital syphyllis;
Grossly, the placenta looks;
1) Hydropic
a. Pale yellow,
b. Waxy, and
c. Enlarged).
2) Polyhydramnios- Because hydramnios is frequently associated with symptomatic congenital infection, fetuses are ultrasonographically followed throughout pregnancy.
Because the antibodies from the maternal compartment are of the immunoglobulin (Ig)G class, they freely cross the placenta, giving most neonates a reactive serologic test if the mother's test was reactive. With symptomatic neonatal infection, often the cord blood serologic test will be higher in titer than the maternal test. No clinically reliable neonatal IgM serologic test is available. Other diagnostic aids include long-bone survey and lumbar puncture, which may help diagnose asymptomatic systemic infection requiring more intense therapy.
A child with congenital syphyllis;
The newborn may have
1) Lymphadenitis.
2) Hepatomegally
3) Splenomegally.
4) The bones usually reveal signs of osteochondritis and an irregular epiphyseal juncture on x-ray.
5) The eyes, central nervous system structures, and other organs may reveal abnormalities at birth, or defects may develop later in untreated cases.
Any infant with the stigmata of syphilis should be placed in isolation until a definite diagnosis can be made and treatment administered.
Newborns with congenital syphilis may appear healthy at birth but often develop symptoms weeks or months later. Examine the body for stigmata of syphilis at intervals of 3 weeks to 4 months. If the mother's serologic test is positive at delivery, the baby's test will also be positive. Obtain serial quantitative serologic tests of the infant's blood for 4 months. A rising titer indicates congenital syphilis, and treatment is indicated.
Click here to access Unit one Content..
Topic 1.6: Syphyllis (cont'd)
5. Latent Syphilis
1. History or serologic evidence of previous infection.
2. Absence of lesions.
3. Serologic test usually reactive; titer may be low.
Pathology of syphyllis;
Syphilis is caused by T pallidum, which is transmitted by direct contact with an infectious moist lesion.
Treponemes pass through intact mucous membranes or abraded skin.
Pathological evolution and history of infection;
1. Ten to 90 days after the treponemes enter;
A primary lesion (chancre) develops. The chancre persists for 1–5 weeks and then heals spontaneously, but it may persist with signs of secondary disease. Serologic tests for syphilis are usually nonreactive when the chancre first appears but become reactive 1–4 weeks later.
2. Two weeks to 6 months (average, 6 weeks)
After the primary lesion appears, the generalized cutaneous eruption of secondary syphilis may appear. The skin lesions heal spontaneously in 2–6 weeks. Serologic tests are almost always positive during the secondary phase.
3. Latent syphilis;
May follow the secondary stage and may last a lifetime, or tertiary syphilis may develop. The latter usually becomes manifest 4–20 or more years after disappearance of the primary lesion.
Untreated syphyllis/chronic complications;
In one-third of untreated cases, the destructive lesions of late (tertiary) syphilis develop. These involve
1) Skin or bone (gummas),
2) The cardiovascular system (aortic aneurysm or insufficiency),
3) The nervous system (meningitis, tabes dorsalis, paresis).
The complications of tertiary syphilis are fatal in almost one-fourth of cases, but one-fourth never show any ill effects.
Laboratory Findings;
1. Identification of the Organism
1) Dark-field examination of specimens from cutaneous lesions, but the recovery period of the treponeme is brief; in most cases, diagnosis depends on the history and serologic tests.
2) An immunofluorescent technique is now available for dried smears.
3) Silver staining for T pallidum of biopsy specimens, placental sections, or autopsy material may confirm the diagnosis in difficult cases. Motile spirochetes can be identified in amniotic fluid obtained transabdominally in women with syphilis and fetal death.
4) PCR is extremely specific for detection of T pallidum in amniotic fluid and neonatal serum and spinal fluid.
5) Newer techniques involving molecular methods are now being used to diagnosis early syphilis. Multiplex PCR is such an assay that can simultaneously detect T pallidum, H ducreyi, and herpes simplex but is not yet commercially available.
2. Serologic Tests
Diagnostic tests after the primary or secondary moist lesion has disappeared are confined largely to serologic testing. Serologic tests become positive several weeks after the primary lesion appears.
1) Nontreponemal Tests;
Nontreponemal tests currently in use are flocculation procedures that include the;
a. VDRL slide test,
b. Rapid reagin test(RPR-Rapid Plasminogen Reagen).
c. Automated reagin test for screening procedures in the field. The latter tests are more sensitive but less specific than the VDRL.
If they are positive, the activity should be verified, and the degree of reactivity should be checked by the VDRL test. Complement fixation tests (eg, Kolmer) are no longer considered usefull.
The VDRL test (the nontreponemal test in widest use) generally becomes positive 3–6 weeks after infection, or 2–3 weeks after the appearance of the primary lesion; it is almost invariably positive in the secondary stage.
The VDRL titer is usually high in secondary syphilis and tends to be lower or even nil in late forms of syphilis, although this is highly variable. A 4-fold falling titer in treated early syphilis or a falling or stable titer in latent or late syphilis indicates satisfactory therapeutic progress.
False-positive serologic reactions are frequently encountered in a wide variety of situations, including;
I. Collagen diseases,
II. Infectious mononucleosis,
III. Malaria,
IV. Febrile diseases,
V. Leprosy,
VI. Vaccination,
VII. Drug addiction,
VIII. Old age,
IX. Pregnancy.
False-positive reactions are usually of low titer and transient and may be distinguished from true-positive results by specific treponemal antibody tests.
3. Treponemal Antibody Tests;
i. FTA-ABS test and
ii. Microhemagglutination assay for Treponema pallidum (MHA-TP.
These detect antibody against Treponema spirochetes.
Both tests are more sensitive and specific than nontreponemal tests (except the MHA-TP test with primary disease.
These tests remain positive despite therapy, so they are not given in titers or used to follow serologic response to treatment.
Differential Diagnosis of syphyllis;
Primary syphyllis;
1. Chancroid,
2. Granuloma inguinale,
3. Lymphogranuloma venereum,
4. Herpes genitalis,
5. Carcinoma,
6. Scabies,
7. Trauma,
8. Lichen planus,
9. Psoriasis,
10. Drug eruption,
11. Aphthosis,
12. Mycotic infections,
13. Reiter's syndrome, and
14. Bowen's disease.
Secondary syphyllis;
1. Pityriasis rosea,
2. Psoriasis,
3. Lichen planus,
4. Tinea versicolor,
5. Drug eruption,
6. Parasitic infections,
7. Iritis,
8. Neuroretinitis,
9. Condyloma acuminatum,
10. Acute exanthems,
11. Infectious mononucleosis,
12. Alopecia, and
13. Sarcoidosis.
Click here to access Unit one Content..
Topic 1.6: Syphyllis (Cont'd)
Prevention;
1) 4Cs.
2) If the patient is known to have been exposed to syphilis, do not wait for the disease to develop to the clinical or reactive serologic stage before giving preventive treatment. Even so, every effort should be made to reach a diagnosis, including a complete physical examination, before administering preventive treatment. It is recommended that any patient who is exposed and becomes symptomatic within 90 days of sexual contact and is seronegative should still be treated. Also, if the exposure occurred more than 90 days earlier and seroconversion takes place, the exposed person should be treated.
3) If the duration since exposure is unknown and the nontreponemal antibody titer is greater than 1:32, treatment is indicated.
4) All pregnant women should undergo a routine serologic test for syphilis at the first visit. The test should be repeated between 28 and 32 weeks' gestation in high-risk regions. If the test result is positive, attention must be given to the patient's prior serologic test and therapy (if any) for syphilis. If doubt exists as to whether the patient has active syphilis, repeat therapy is far better than the risk of congenital syphilis.
Rx of Syphyllis;
Early Syphilis and Contacts
Primary, secondary, and early latent syphilis (<1 year's duration):
1. Benzathine penicillin G 2.4 million units IM once.
2. Tetracycline hydrochloride 500 mg QID X 2/52
3. Doxycycline 100mg BD X 2/52.
4. For nonpregnant penicillin-allergic patients. Erythromycin estolate should not be administered to pregnant women because of potential drug-related hepatotoxicity.
Ceftriaxone 1 g daily IM or IV for 8–10 days may be effective, but data on this regimen are limited.
Late Syphilis
Includes latent syphilis of indeterminate duration or more than 1 year's duration, except neurosyphilis.
1. Benzathine penicillin G 2.4 million units IM weekly for 3 successive weeks (7.2 million units total).
2. Tetracycline hydrochloride 500 mg orally QID X 2/52.
3. Doxycycline 100mg BD X 2/52.
Rx of Syphilis in Pregnancy;
Treat as indicated above, except that tetracycline or erythromycin is not recommended.
If serologic tests are equivocal (eg, possible biologic false-positive result), it is better to err on the side of early treatment.
Because of the increased risk for treatment failure, a second dose of 2.4 million units of penicillin IM is often recommended.
Penicillin-allergic patients can be given oral desensitization therapy using gradually larger doses of phenoxymethyl penicillin suspension to achieve a temporary tolerant state that allows parenteral penicillin therapy. This is particularly useful in circumventing compliance problems due to hyperemesis or drug-induced gastric upset.
Rx of Congenital Syphilis;
Adequate maternal treatment before 16–18 weeks' gestation prevents congenital syphilis. Treatment thereafter may arrest fetal syphilitic infection, but some stigmata may remain.
1. Benzathine penicillin G 50,000 U/kg IM as a single injection, for asymptomatic infants without neurosyphilis.
2. Aqueous crystalline penicillin G 50,000 U/kg IV every 8–12 hours, or procaine penicillin G 50,000 U/kg IM once daily for 10–14 days, for symptomatic infants or those with neurosyphilis.
Jarisch-Herxheimer Reaction in syphyllis;
A febrile reaction may occur in 50–75% of patients with early syphilis treated with penicillin.
This occurs 4–12 hours after injection and is completed by 24 hours. Its cause is uncertain but may involve a release of treponemal toxic products upon organism lysis. The reaction is generally benign but may trigger labor or fetal distress. Prophylaxis with antipyretics or corticosteroids is of unknown value.
Syphyllis in HIV;
No specific changes in treatment are currently necessary, but close follow-up is necessary to ensure adequate treatment.
Recommendations include serology tests every 3 months for 1 year and twice during the second year.
Click here to access Unit one Content..
Topic 1.7: Vaginitis
This is generally inflammation of the vagina.
Vaginitis is a clinical syndrome characterized by;
i. Vaginal discharge,
ii. Vulvar irritation, or
iii. Malodorous discharge.
Classification of vaginitis;
This is often broken down into 2 entities:
1) Infectious vaginitis and
2) Atrophic vaginitis.
Infectious vaginitis;
Infectious vaginitis is most frequently caused by 1 of 3 diseases:
1. Trichomoniasis,
2. Bacterial vaginosis, or
3. Candidiasis.
Bacterial Vaginosis (Corynebacterium vaginale Vaginitis; Gardnerella vaginalis Vaginitis)
Although bacterial vaginosis is the most prevalent vaginal infection, almost 50% of affected women are asymptomatic.
The term bacterial vaginosis refers to the intricate changes of vaginal bacterial flora with a loss of lactobacilli, an increase in vaginal pH (pH > 4.5), and an increase in multiple anaerobic and aerobic bacteria.
Clinical criteria for diagnosing bacterial vaginosis;
i. Homogeneous white, noninflammatory discharge;
ii. Microscopic presence of > 20% clue cells;
iii. Vaginal discharge with pH > 4.5; and
iv. Fishy odor with or without addition of 10% potassium hydroxide (KOH).
The pathogen in bacterial vaginosis;
Gardnerella vaginalis (formerly designated Corynebacterium vaginale and Haemophilus vaginalis) is a small, nonmotile, nonencapsulated, pleomorphic rod that stains variably with Gram's stain.
It may be spread by sexual contact and, although of low virulence, causes vaginitis.
The disorder may be atypical and even more troublesome when G vaginalis coexists with more virulent organisms.
G vaginalis is not the only cause of bacterial vaginosis.
Fishy odour in bacterial vaginosis;
The characteristic fishy odor of bacterial vaginosis is due to anaerobic bacteria, such as;
1. Bacteroides,
2. Prevotella,
3. Peptostreptococcus, and
4. Mobiluncus spp., and
5. Genital mycoplasmas.
G vaginalis infection is often overlooked. It may be suspected on the basis of the microscopic appearance of unstained exfoliated vaginal cells in a wet preparation that appears to be dusted with many small dark particles, actually G vaginalis organisms.
These "clue cells" are presumptive evidence of the presence of this organism. In case of mixed infection (eg, with Candida albicans), it may not be possible to make the diagnosis except by culture.
Gram's stain is another method useful for making the diagnosis of bacterial vaginosis.
Bacterial vaginosis and adverse pregnancy outcome;
Observational studies have consistently shown an association between bacterial vaginosis and adverse pregnancy outcomes, including;
1) Preterm delivery,
2) Preterm premature rupture of membranes,
3) Spontaneous abortion, and
4) Preterm labor.
However, 2 large randomized, placebo-controlled trials demonstrated that treatment of bacterial vaginosis in asymptomatic, pregnant women with metronidazole does not prevent preterm deliveries.
The CDC recommends that pregnant women with a history of preterm delivery and asymptomatic bacterial vaginosis be evaluated for treatment.
Rx of bacterial vaginosis;
1. Oral metronidazole 500 mg BD X 1/52.
2. Antibacterial gels/creams
a. Clindamycin cream 2%, 1 applicatorful (5 g) intravaginally at night for 7 days; and
b. Metronidazole gel 0.75%, 1 applicatorful (5 g) intravaginally once daily for 5 days.
3. Oral metronidazole 2 g in a single dose;
4. Clindamycin 300 mg BD X 1/52
5. Clindamycin ovules 100 g intravaginally once at bedtime for 3 days.
Four randomized controlled trials have demonstrated overall cure rates of 95% for the 7-day metronidazole regimen and 84% for the single 2-g regimen.
During pregnancy, oral treatment is preferred to local agents to ensure adequate tissue levels of the bactericidal drug. The recommended regimen is metronidazole 250 mg orally 3 times daily for 7 days or clindamycin 300 mg orally twice daily for 7 days.
Click here to access Unit one Content..
Topic 1.8: Urethritis and cervicitis
Aetiology;
Urethral mucopurulent or purulent discharge is commonly caused by
1. Neisseria gonorrhoeae,
2. C trachomatis, or
3. Genital herpes.
Although asymptomatic infections are common, some patients experience slow-onset dysuria with vaginal discharge and/or irregular bleeding.
Urethritis and cervicitis are often coinfections. Both are reportable STDs, and clinicians must mandate that partners of patients obtain diagnostic and therapeutic interventions.
Cervicitis is an inflammation of either the ectocervical cells or the glandular cells composing the cervical epithelium.
The ectocervical squamous cells are contiguous with the vaginal epithelium and can be infected by the same organisms that cause inflammatory vaginitis. The glandular cells of the endocervix are more commonly inflamed by N gonorrhoeae and C trachomatis.
1) Gonorrhea;
Practical essentials;
i. Most affected women are asymptomatic carriers.
ii. Purulent vaginal discharge.
iii. Urinary frequency and dysuria.
iv. May progress to pelvic infection or disseminated infection.
Aetiology of gonorrhea;
Neisseria gonorrhoeae.
This is a gram-negative diplococcus that forms oxidase-positive colonies and ferments glucose.
The organism may be recovered from the urethra, cervix, anal canal, or pharynx. It causes ascending infections.
N gonorrhoeae is rapidly killed by drying, sunlight, heat, and most disinfectants.
The columnar and transitional epithelium of the genitourinary tract is the principal site of invasion. The organism may enter the upper reproductive tract, causing salpingitis with its attendant complications.
It has been estimated that after exposure to an infected partner, 20–50% of men and 60–90% of women become infected.
Without therapy, 10–17% of women with gonorrhea develop pelvic infection.
Depending on the geographic location and population involved, N gonorrhoeae is often present with other STDs.
Traditionally, women with gonorrhea are considered to be at risk for incubating syphilis. It has been shown that 20–40% also have Chlamydia infection.
Clinical Features of gonorrhea;
1. Early Symptoms
1) Most women with gonorrhea are asymptomatic.
2) Vaginal discharge,
3) Urinary frequency or dysuria, and
4) Rectal discomfort.
The incubation period is only 3–5 days.
The vulva, vagina, cervix, and urethra may be inflamed and may itch or burn. Specimens of discharge from the cervix, urethra, and anus should be taken for culture from the symptomatic patient.
A stain of purulent urethral exudate may demonstrate gram-negative diplococci in leukocytes. Similar findings in a purulent cervical discharge are less conclusively diagnostic of N gonorrhoeae.
Gonorrhoea Bartholinitis/Bartholins abscess;
Unilateral swelling in the inferior lateral portion of the introitus suggests involvement of Bartholin's duct and gland.
In early gonococcal infections, the organism may be recovered by gently squeezing the gland and expressing pus from the duct.
Enlargement, tenderness, and fluctuation may develop, signifying abscess formation. N gonorrhoeae is then less frequently recovered; however, the prevalence of infection with other bacteria merits a search for these pathogens. Spontaneous evacuation of pus often occurs if the incision is not drained. The infection may result in asymptomatic cyst formation
Gonorrhoea Anorectal Inflammation/Proctitis;
i. Anal itching,
ii. Anal pain,
iii. Anal discharge, or
iv. Bleeding occurs rarely.
Most women are asymptomatic and acquire infection by perineal spread of vaginal secretions rather than by anal intercourse
Gonorrhoea Pharyngitis;
Acute pharyngitis and tonsillitis rarely occur; most infections are asymptomatic.
Disseminated gonococcal Infection;
For unknown reasons, asymptomatic carriers can develop systemic infection.
Commonly, a triad of
i. Polyarthralgia,
ii. Tenosynovitis, and
iii. Dermatitis is seen, or
iv. Purulent arthritis without dermatitis.
Septicemia is more common in the former clinical setting and N gonorrhoeae cultured from joint aspirates in the latter. Endocarditis and meningitis have been described.
Gonococcal Conjunctivitis;
In adults, ophthalmic infection is usually due to autoinoculation.
Ophthalmia neonatorum may result from delivery through an infected birth canal.
Gonococcal Vulvovaginitis in Children;
Gonococcal invasion of nonkeratinized membranes in prepubertal girls produces severe vulvovaginitis.
The typical sign is a purulent vaginal discharge with dysuria.
The genital mucous membranes are red and swollen. Infection is commonly introduced by adults, and in such cases the physician must consider the possibility of child abuse.
Laboratory Dx of Gonorrhoea;
A presumptive diagnosis of gonorrhea can be made based on examination of the stained smear; however, confirmation requires positive identification on selective media.
1. Secretions are examined under oil immersion for presumptive identification which identifies Gram-negative diplococci.
2. Culture which yield gram negative diplococci that are oxidase-positive and obtained from selective media (Thayer-Martin or Transgrow) usually signify N gonorrhoeae.
3. Carbohydrate fermentation tests may be performed, but in addition to being time-consuming and expensive, they occasionally yield other species of Neisseria. Therefore, cultures are reported as "presumptive for N gonorrhoeae."
4. Chlamydial cultures or direct smear testing (ELISA or immunofluorescent staining) of the cervix and a serologic test for syphilis should also be obtained.
5. Other techniques for detecting gonorrhea include enzyme immunoassay from cervical swab or urine specimens, DNA probes from endocervical swabs, and nucleic acid amplification tests (NAATs) from endocervical swabs, liquid Papanicolaou (Pap) specimens, vaginal swabs, and urine specimens.
Complications of Gonorrhoea;
1) Salpingitis and the complications that may arise from salpingitis (Salpingitis-Peritonitis).
2) Cervicitis- gonorrhoeae can be recovered from the cervix in approximately 50% of women with salpingitis.
3) It is important to note that asymptomatic carriers can also develop tubal scarring, infertility, and increased risk for ectopic gestations.
Rx of gonorrhoea
Any patient with gonorrhea must be suspected of having other STDs (eg, syphilis, HIV, and chlamydial infection) and managed accordingly.
Treatment should cover N gonorrhoeae, C trachomatis, and incubating syphilis.
Dual therapy has contributed greatly to the declining prevalence of chlamydial infections.
Therefore, if chlamydial infection is not ruled out, the following regimens should be given with doxycycline or azithromycin.
a) Adults/Non pregnant;
1) Ceftriaxone 125 mg IM once, plus doxycycline 100 mg orally twice daily for 7 days
2) Azithromycin 1 g orally in a single dose if chlamydial infection is not ruled out;
3) Cefixime 400mg orally once, plus doxycycline or azithromycin as above;
4) Ofloxacin 400mg,
5) Levofloxacin 250 mg,
6) Ciprofloxacin 500mg orally once in nonpregnant, nonlactating patients over 17 years old, plus doxycycline or azithromycin as above.
b) Pregnancy;
Pregnant women should not be treated with quinolones or tetracyclines.
They should be treated with a recommended or alternate cephalosporin.
If cephalosporins are not tolerated, spectinomycin 2 g IM should be given along with treatment for diagnosed or presumptive C trachomatis.
The incidence of penicillinase-producing strains of N gonorrhoeae (PPNGs) is increasing and is spreading from coastal areas to the center of the United States. They are unresponsive to previously recommended conventional therapy such as penicillin, ampicillin, or amoxicillin. Currently recommended cephalosporins and quinolones and regimens with -lactamase inhibitors are effective therapy against PPNG strains.
c) Rx of disseminated gonococcal infections;
Disseminated gonococcal infection should be treated in the hospital initially.
Evidence for endocarditis or meningitis should be sought.
Recommended regimens include;
i. Ceftriaxone 1 g IM or IV every 24 hours,
ii. Cefotaxime or ceftizoxime 1 g IV every 8 hours.
iii. For patients with -lactamase allergy, spectinomycin 2 g IM every 12 hours can be used. Testing for chlamydia should be performed or therapy given.
d) Rx of gonorrhea in Neonates and Children;
Infants born to women with untreated gonorrhea should be treated with
· Ceftriaxone 25–50 mg/kg IV or IM, not to exceed 125 mg. It should be given cautiously to premature or hyperbilirubinemic infants.
Prognosis of Syphyllis;
The prognosis is excellent for patients with gonorrhea who receive prompt treatment.
Infertility may result from even a single episode.
Click here to access Unit one Content..
Topic 1.9: Chlamydia
Practice essentials;
Genital infection with this organism is the most common sexually transmitted bacterial disease in women.
Chlamydiae are obligate intracellular microorganisms that have a cell wall similar to that of gram-negative bacteria.
They are classified as bacteria and contain both DNA and RNA. They divide by binary fission, but like viruses they grow intracellularly.
They can be grown only by tissue culture. With the exception of the L serotypes, chlamydiae attach only to columnar epithelial cells without deep tissue invasion. As a result of this characteristic, clinical infection may not be apparent.
For example, infections of the eye, respiratory tract, or genital tract are accompanied by discharge, swelling, erythema, and pain localized to these areas only. C trachomatis infections are associated with many adverse sequelae due to chronic inflammatory changes as well as fibrosis (eg, tubal infertility and ectopic pregnancy).
The proposed mechanism for the pathogenesis of chlamydial disease is an immune-mediated response. This mechanism has been supported C trachomatis vaccine studies in humans and monkeys as well as other animal model studies.
Evidence indicates that a 57-kDa chlamydial protein, which is a member of 60-kDa heat shock proteins, plays a role in the immunopathogenesis of chlamydial disease.
Risk factors for chlamydia;
Certain factors may be predictive of women with a greater likelihood of acquiring C trachomatis.
1. Sexually active women younger than 20 years have chlamydial infection rates 2–3 times higher than the rates of older women.
2. The number of sexual partners .
3. Lower socioeconomic status are associated with higher chlamydial infection rates.
4. Persons who use barrier contraception are less frequently infected by C trachomatis than are those who use no contraception.
5. Women who use oral contraceptives may have a higher incidence of cervical infection than women not using oral contraceptives.
Cervical infection in pregnant women varies from 2–24% and is most prevalent in young, unmarried women of lower socioeconomic status in inner-city environments.
Screening for chlamydia;
The CDC recommends screening sexually active adolescent girls at their routine yearly gynecologic examination, as well as women 20–24 years old, especially those who have new or multiple partners, and those who inconsistently use barrier contraceptives.
Clinical features of chlamydial infections;
1. Women with chlamydial infection not uncommonly are asymptomatic.
2. Women with cervical infection generally have a mucopurulent discharge with hypertrophic cervical inflammation. Salpingitis may be unassociated with symptoms.
Laboratory Dx of Chlamydia;
The diagnosis of chlamydial infection is based solely on laboratory tests.
1) Cell culture isolation has a sensitivity of 70–90%; however, this specialized modality is not widely available. Cell culture is the detection method of greatest specificity (almost 100%), but the cost can be prohibitive, and a 3- to 7-day delay in diagnosis is required.
2) Serologic methods, either the complement fixation or microimmunofluorescence test, are not totally accurate, as 20–40% of sexually active women have positive antibody titers. In fact, most women with microimmunofluorescent antibody do not have a current infection.
3) Direct smear fluorescent antibody testing requires a fluorescence microscope, and processing time is only 30–40 minutes. Sensitivity is 90% or higher, with a specificity of 98% or higher if an experienced microscopist and a satisfactory specimen are available. This test appears to be the most promising, and when tissue samples (endometrial or uterine tube) are being evaluated, it has been reported to be more accurate.
4) PCR, ligase chain reaction, and current DNA probes used for detection of C trachomatis may be more rapid and less expensive. Nucleic acid hybridization methods (DNA probe) require only 2–3 hours of processing time. The DNA probe assay is specific for C trachomatis; cross-reactivity with C pneumoniae and C psittaci has not been reported. To ensure high specificity, a competitive probe assay has been produced and is currently being evaluated in clinical trials. Recent reports indicate PCR positivity with negative culture. PCR may be the most sensitive and specific test method for chlamydia.
Differential Diagnosis of Chlamydia;
i. Mucopurulent cervicitis.
ii. Salpingitis.
Complications of chlamydia;
1) Infertility due to tubal obstruction and ectopic pregnancy, are the most dire complications of these infections.
2) Pregnant women with cervical chlamydial infection can transmit infections to their newborns; evidence indicates that up to 50% of infants born to such mothers will have inclusion conjunctivitis.
3) In perhaps 10% of infants, an indolent chlamydial pneumonitis develops at 2–3 months of age.
4) This pathogen may cause otitis media in the neonate.
5) Whether maternal cervical infection with Chlamydia causes significantly increased fetal and perinatal wastage by abortion, premature delivery, or stillbirth is uncertain.
Treatment of Chlamydia;
1. Doxycycline 100 mg BD X 1/52.
2. Azithromycin 1 g orally as a single dose.
3. Erythromycin 500 mg QID X 1/52.
4. Patients who cannot tolerate erythromycin should consider ofloxacin 300 mg twice daily or levofloxacin 500 mg orally once daily for 7 days.
5. Administration of high doses of ampicillin has resulted in elimination of C trachomatis from the cervices of women with acute salpingitis. Addition of the irreversible -lactamase enzyme inhibitor sulbactam increases in vitro antichlamydial activity.
Chlamydia in pregnancy;
Pregnant women are advised to take
1. Erythromycin base 500 mg QID X 1/52.
2. Amoxicillin 500 mg TDS X 1/52.
3. Azithromycin 1.5 g STAT.
Click here to access Unit one Content..
SUMMARY OF STIs AND SYNDROMIC MANAGEMENT
Reference Material
1. Harrison's Principles of internal medicine 17th edition.
2. Davidson's Principles and Practice of medicine, 21st Edition.
3. Tropical Diseases AMREF
4. Kumar and Clerk Text book of clinical Medicine 6E Edition
5. Oxford Textbook of Medicine Michael Glynn, William Drake, Clinical Methods, 23rd Edition, 2012, London UK
Click here to access Unit one Content..
Topic One: Further Reading
Reference Material
1. Harrison's Principles of internal medicine 19th edition.
2. Davidson's Principles and Practice of medicine, 21st Edition.
3. Tropical Diseases AMREF
4. Kumar and Clerk Text book of clinical Medicine 6E Edition
5. Oxford Textbook of Medicine Michael Glynn, William Drake, Clinical Methods, 23rd Edition, 2012, London UK
Click here to access Unit one Content..
Topic 2.1: HIV/AIDS
INTRODUCTION TO IMMUNE DEFICIENCY SYNDROMS;
Immune deficiency is a state where the body’s immune system is unable to detect pathologies, antigens and disorders that cause disease and thus provide adequate protection to cells and tissues from damage caused by the action of these disease process or pathogens.
Aetiology of immune deficiency;
- Inherited causes;
1) Sickle cell disease- Occur in splenic sequestration and asplenism that follows the condition.
2) Leukemia;- Results to the destruction of haemopoietic stem cells resulting to pancytopenia (reduction in all cell types) including leucocytes, neutrophils and eosinophils.
- Acquired;
1) Infections;
i. Bacterial.
1. Tuberculosis.
ii. Viral.
1. HIV.
2. CMV
3. EBV.
4. Yellow fever.
iii. Cancers.
2) Noninfectious;
i. Nutritional
1. Protein energy malnutrition.
2. Starvation.
ii. Drugs;
1. Sulphonamides.
2. Chemotherapy for cancer.
3. Corticosteroids.
iii. Irradiation of the PHSC.
Acquired agents cause acquired immune deficiency syndrome (AIDS) while inherited factors cause inherited immune deficiency syndrome (IIDS).
The most common infectious agent that cause immune deficiency in our setting is HIV. Cancers are catching up as a significant cause of immune suppression.
Click here to access Unit one Content..
Topic 2.2: HIV/AIDS
INTRODUCTION TO IMMUNE DEFICIENCY SYNDROMS;
Immune deficiency is a state where the body’s immune system is unable to detect pathologies, antigens and disorders that cause disease and thus provide adequate protection to cells and tissues from damage caused by the action of these disease process or pathogens.
Aetiology of immune deficiency;
- Inherited causes;
1) Sickle cell disease- Occur in splenic sequestration and asplenism that follows the condition.
2) Leukemia;- Results to the destruction of haemopoietic stem cells resulting to pancytopenia (reduction in all cell types) including leucocytes, neutrophils and eosinophils.
- Acquired;
1) Infections;
i. Bacterial.
1. Tuberculosis.
ii. Viral.
1. HIV.
2. CMV
3. EBV.
4. Yellow fever.
iii. Cancers.
2) Noninfectious;
i. Nutritional
1. Protein energy malnutrition.
2. Starvation.
ii. Drugs;
1. Sulphonamides.
2. Chemotherapy for cancer.
3. Corticosteroids.
iii. Irradiation of the PHSC.
Acquired agents cause acquired immune deficiency syndrome (AIDS) while inherited factors cause inherited immune deficiency syndrome (IIDS).
The most common infectious agent that cause immune deficiency in our setting is HIV. Cancers are catching up as a significant cause of immune suppression.
Click here to access Unit one Content..
Topic 2.2: HIV/AIDS (Cont'd)
HIV/AIDS;
HIV disease is a chronic infectious disease caused by the Human Immuno Deficiency Virus, which is characterized by spectrum starting from primary infection, with or without the acute syndrome, followed by a relatively long period of asymptomatic stage after which in most patient’s progress to advanced and life threatening disease (AIDS).
Historical Back Ground
1981: AIDS was first recognized in USA among Homosexual males PCP was seen among 5 homosexuals. Kaposi’s sarcoma was diagnosed in 26 homosexuals.
1983: HIV virus was isolated from a patient with lymphadenopathy
1984: HIV virus was clearly demonstrated to be the causative agent for AIDS
Mode of Transmission of HIV;
1. Sexual Transmission:
Is the major mode of transmission worldwide ( 90 % )
1) The virus is found in high quantities in the sexual fluids (seminal and vaginal fluid) of people with HIV infection with in infected monocytes and in cell-free state.
2) Anal sex appears to be the sexual practice carrying the highest risk of transmitting HIV. The reasons being rectal mucosa is thin and fragile and there are susceptible cells ( Langerhans cells ) in the rectal mucosa.
3) Vaginal sex is also an effective from of transmission.
4) The presence of other STDs like Syphilis , Gonorrhea etc increase the risk of acquiring or transmitting HIV infection by several fold, as the quantity of the virus in seminal or vaginal fluid significantly increase and the number of infected monocytes is high around the genital area in patients with STDs.
5) Oral sex may have some risk however there are no reports so far attributable to oral sex.
2. Transmission through blood and blood products;
IV drug abusers who share needles and syringes have high risk .This is the main mode of transmission in Eastern Europe.
Blood or blood products transfusion from infected donors (the risk of infection I s90-100 %). Currently the risk is very minimal as blood and blood products are screened carefully using antibody and p24 antigen testing to identify donors in the widow period.
Transmission through sharp instruments, injection needles etc. There may be a risk of transmission from one patient to another or from an infected patient to health care provider
3. Mother to Child Transmission;
Without any intervention, the risk of mother to child transmission is 30-45% in the developing world and 15-30% in the developed world .
HIV may be transmitted from infected mothers to children during;
i. Pregnancy:-10 % before the 3rd Trimester
ii. Labor and Delivery:- 70 % late pregnancy and during labor
iii. Breast Feeding :- 10-15 %
MTCT is by far the largest source of HIV infection in children under 15.
FACTORS INFLUENCING MTCT
1. Maternal Factors;
i. Maternal viral Load: The higher the viral load, the higher is the risk of MTCT
ii. Woman becomes infected with HIV during pregnancy
iii. Severe immune deficiency
iv. Clinical and immunological state
v. Use of ART during pregnancy and postpartum to mother and newborn
vi. Viral, bacterial, and parasitic placental infection (especially malaria)
vii. Nutritional status, particularly vitamin A
2. Labor and Delivery;
i. Prolonged rupture of membranes (>4 hours)
ii. Acute chorioamnionitis
iii. Invasive fetal monitoring or delivery techniques
iv. Mixing of maternal and fetal body fluids
v. Episiotomy
vi. First infant in a multiple birth
3. Fetal Conditions;
i. Premature delivery
ii. Immature immune status
iii. First-born twin
4. Infant Feeding Practices/Breastfeeding;
i. Mother becomes infected with HIV while breastfeeding (risk increases up to 20%)
ii. Mixed feeding (breast milk and other foods) increases risk
iii. Breast pathologies (lesions, infections)
iv. Advanced disease in the mother
v. Poor maternal nutritional status
vi. Breastfeeding during the first 4–6 months
vii. The longer mother breastfeeds, the greater the risk
HIV is not spread via non-sexual everyday casual contact between people like kissing, hugging and sharing common utensils, baths etc.
HIV infected people are considered most infectious soon after acquiring the infection and during the AIDS (symptomatic) phase. However, remember that it is possible to transmit HIV any time during the disease.
Click here to access Unit one Content..
Topic 2.3: HIV/AIDS (Structure and Lifecycle of HIV)
The pathogen- HIV;
HIV is a retrovirus which belongs to the sub family of lente virus. HIV virus is cytopathic virus.
There are 2 main Types of the virus;
1) HIV- 1:
This is the most common cause of HIV Disease throughout the world and it has several groups and subtypes;
i. M group which comprises 9 subtypes: A,B,C,D,F,G,H,J,K, as well as growing number of major circulating recombinant forms ( CRFs ) e.g. AE , AG etc
ii. G roup O (outliers): relatively rare seen in Cameron and Gambia
iii. N group: (reported only in Cameroon)
Global Pattern of HIV -1 distribution
1. Africa: > 75 % of strains recovered to date have been subtypes A, C and D with C being the most common
2. Europe and Americas: subtype B is the predominant strain.
3. Asia: recombinant forms such AE account of the infections in south East Asia while subtype C is prevalent in India. Subtype B is also seen in Asia.
HIV - 2:
Was first identified in West Africa and it is mostly confined to West Africa, however a number of cases which can be traced to West Africa are found worldwide.
Viral Morphology;
HIV is Spherical shaped virus. The most important parts of the virus are:-
1) Its viral envelop has many small spikes which consists of two important glycopropteins;
i. Glycoprotein- gp41 and
ii. Glycoprotein-gp120
Which play an important role when the virus attaches to its host cells.
2) The viral capsid ( core ) which contains two single stranded viral RNA and an important enzyme for the virus called reverse transcriptase enzyme.
3) The reverse transcriptase enzyme plays an important step in the life cycle of the virus. It converts the single stranded viral RNA into double stranded DNA ( this process is called reverse transcription).
Characteristics of HIV;
HIV infect cells that express CD4 receptor molecules;
1. The CD4 receptors are present on various types of blood cells including
i. Lymphocytes,
ii. Macrophages,
iii.
Monocytes,
iv. Tissue cells (e.g. Dendirtic cells in the genital tract and ano-rectal region) and
v. Glial cells of brain.
2. Successful entry of the virus to a target cell also requires cellular co-receptors
i. A fusion co-receptor is designated;
a. CXCR5 for T-cell tropic stain.
b. CCR4 for monocyte -macrophage tropic strains.
c. The receptor and co-receptors of CD4 cells interact with HIV’s gp-120 and gp-41 proteins during entry into a cell.
Life Cycle of HIV: Replication;
1. Attachment /binding and fusion of the virus to the host cells
The receptor and co-receptors of CD4 cells interact with HIV’s gp-120 and gp-41 proteins during entry into a cell.
2. Uncoatting of the viral capsid and release of Viral RNA into the cytoplasm of the host cell.
3. Reverse transcription: Viral RNA is concerted in to Double stranded DNA by reverse transcriptase enzyme
4. Translocation : viral DNA is Imported to cell nucleus
5. Integration of proviral DNA to host-cell DNA
6. Cellular activation causes transcription (copying) of HIV DNA back to RNA
i. Some RNA translated to HIV proteins.
ii. Other RNA moved to cell membrane
7. Viral Assembly :HIV assembled under cell membrane and buddes from cell
8. Maturation
: viral Proteases enzymes cleave longer proteins in to important viral proteins
and help to convert immature viral particle into and infectious HIV.
Click here to access Unit one Content..
Topic 2.4: HIV/AIDS (Pathogenesis of HIV)
Pathogenesis of HIV/AIDS;
CD4 positive T-Lymphocytes (also known as T helper cells) play central role in the defense mechanism of the body against infection. They mainly coordinate the Cell mediated immune system and also assist the antibody mediated immune system.
1. HIV virus has special affinity to CD4 T-cells and infects them.
2. HIV infection is characterized by a profound immunodeficiency from progressive decline of T-helper cells.
3. The pathogenetic mechanism of HIV disease is multi-factorial and multiphasic and it differs in different stage of the disease.
Mechanism of CD4 Cell Depletion on HIV infection;
1. HIV-mediated direct cytopathicity (single cell killing) – infected CD4 cells die.
2. HIV-mediated syncytia formation.
3. Defect in CD4 T-cell regeneration in relation to the rate of destruction.
4. Maintenance of homeostasis of total T-lymphocytes (decreased CD4, increased CD8)
5. HIV-specific immune response (killing of virally infected and innocent cells)
6. Auto-immune mechanism.
7. Programmed cell death (apoptosis).
Qualitative abnormalities (even the existing CD4 cells are dysfunctional);
1) Impaired expression of IL-2.
2) Defective IL-2 and INF-Alfa production.
3) Decreased help to B-cells in production of immunogloblins.
Peculiar Characteristics of HIV and reasons for Persistent Viremia;
HIV is a unique infection in that, though the body reacts by producing antibodies to destroy the virus, the virus is not cleared, except partially in the early period of infection.
A chronic infection is established, and it persists with varying degrees of viral replication. Viral replication is continuous in all stages (early infection, during the long period of clinical latency, and in advanced stage.) There is no virological latency.
Despite robust immune reaction, HIV evades elimination by the immune system and a chronic infection is established. Some of the mechanisms are:
i. High level of viral mutation – HIV has an extraordinary ability to mutate
ii. Large pool of latently infected cells that cannot be eliminated by viral-specific CTLs
iii. Virus homes in lymphoid organs, while antibody is in the circulation
iv. Exhaustion of CD8 T-lymphocytes by excessive antigen stimulation
v. HIV attacks CD-4 T-cells, which are central to both humeral and-cell mediated immunity.
vi. HIV seeds itself in areas of the body where sufficient antibodies might not reach, e.g., the central nervous system.
Progression of HIV is different in different individuals;
1. Rapid progressors:
After the initial infection patients progress fast and develop OIs and die within 2-3 years. Account for 15 % of all patients.
2. Normal Progressors:
After the initial primary infection patients remain health for 6- 8 years before they start having overt clinical manifestations: account for 80 % of all patients.
3. Long term survivors:
Patients who remain alive for 10-15 years after initial infection. In most the diseases might have progressed and there may be evidences of immunodeficiency.
4. Long term non progressors:
This is individuals who have been infected with HIV for > 10 years. Their CD4 count may be in the normal range and they may remain clinically stable for several years
Factors affecting disease progression in HIV Infected individuals;
1) Viral set point: The level of steady-state viremia (set-point) at six months to one year after infection, has an important prognostic implication for progression of HIV disease. Those with a high viral set-point have faster progression to AIDS, if not treated
2) Immune response
i. High CD8 slow progression
ii. Low CD8 rapid decline
3) Viral type; HIV 2 slow course
4) Concomitant conditions
i. Malnutrition hastens the progression of HIV
ii.
Chronic infectious conditions e.g.
Tuberculosis
Click here to access Unit one Content..
Topic 2.5: HIV/AIDS (Diagnosis and Laboratory Monitoring of HIV)
Pathogenesis of HIV/AIDS;
CD4 positive T-Lymphocytes (also known as T helper cells) play central role in the defense mechanism of the body against infection. They mainly coordinate the Cell mediated immune system and also assist the antibody mediated immune system.
1. HIV virus has special affinity to CD4 T-cells and infects them.
2. HIV infection is characterized by a profound immunodeficiency from progressive decline of T-helper cells.
3. The pathogenetic mechanism of HIV disease is multi-factorial and multiphasic and it differs in different stage of the disease.
Mechanism of CD4 Cell Depletion on HIV infection;
1. HIV-mediated direct cytopathicity (single cell killing) – infected CD4 cells die.
2. HIV-mediated syncytia formation.
3. Defect in CD4 T-cell regeneration in relation to the rate of destruction.
4. Maintenance of homeostasis of total T-lymphocytes (decreased CD4, increased CD8)
5. HIV-specific immune response (killing of virally infected and innocent cells)
6. Auto-immune mechanism.
7. Programmed cell death (apoptosis).
Qualitative abnormalities (even the existing CD4 cells are dysfunctional);
1) Impaired expression of IL-2.
2) Defective IL-2 and INF-Alfa production.
3) Decreased help to B-cells in production of immunogloblins.
Peculiar Characteristics of HIV and reasons for Persistent Viremia;
HIV is a unique infection in that, though the body reacts by producing antibodies to destroy the virus, the virus is not cleared, except partially in the early period of infection.
A chronic infection is established, and it persists with varying degrees of viral replication. Viral replication is continuous in all stages (early infection, during the long period of clinical latency, and in advanced stage.) There is no virological latency.
Despite robust immune reaction, HIV evades elimination by the immune system and a chronic infection is established. Some of the mechanisms are:
i. High level of viral mutation – HIV has an extraordinary ability to mutate
ii. Large pool of latently infected cells that cannot be eliminated by viral-specific CTLs
iii. Virus homes in lymphoid organs, while antibody is in the circulation
iv. Exhaustion of CD8 T-lymphocytes by excessive antigen stimulation
v. HIV attacks CD-4 T-cells, which are central to both humeral and-cell mediated immunity.
vi. HIV seeds itself in areas of the body where sufficient antibodies might not reach, e.g., the central nervous system.
Progression of HIV is different in different individuals;
1. Rapid progressors:
After the initial infection patients progress fast and develop OIs and die within 2-3 years. Account for 15 % of all patients.
2. Normal Progressors:
After the initial primary infection patients remain health for 6- 8 years before they start having overt clinical manifestations: account for 80 % of all patients.
3. Long term survivors:
Patients who remain alive for 10-15 years after initial infection. In most the diseases might have progressed and there may be evidences of immunodeficiency.
4. Long term non progressors:
This is individuals who have been infected with HIV for > 10 years. Their CD4 count may be in the normal range and they may remain clinically stable for several years
Factors affecting disease progression in HIV Infected individuals;
1) Viral set point: The level of steady-state viremia (set-point) at six months to one year after infection, has an important prognostic implication for progression of HIV disease. Those with a high viral set-point have faster progression to AIDS, if not treated
2) Immune response
i. High CD8 slow progression
ii. Low CD8 rapid decline
3) Viral type; HIV 2 slow course
4) Concomitant conditions
i. Malnutrition hastens the progression of HIV
ii. Chronic infectious conditions e.g. Tuberculosis
Serologic Tests:
1. HIV antibody tests :-
Detect antibodies formed by the immune system against HIV
1) ELISA : used to be standard screening test for HIV
· Tests for a number of antibody proteins in combination
· A very sensitive test ( . 99.5 % ), but not very specific
· A positive result needs to be confirmed by Western blot for confirmation
· The test need skilled personnel , takes several hours.
2) Western blot: is an excellent confirmatory test.
· It has high specificity but relatively poor sensitivity
· It should not be used for screening purpose
3) Rapid HIV antibody testes
Advantages of HIV Rapid tests:
i. Rapid tests have reasonably good sensitivity and specificity ( >99 % ).
ii. Easy logistically , does not need continuous water or electric supply.
iii. Can be done by less skilled personnel and the interpretation of results is easy.
iv. Test result can be made available in < 30 minutes.
4) HIV antigen assays ( Tests )
1) P24 antigen capture assay:
This test detects p24 viral protein in the blood of HIV infected individuals. This viral protein can be detected during early infection, before seroconversion. Thus this test is used to detect blood donors during the Window period.
2. DNA – PCR : Viral replication
This is an extremely sensitive test -can detect 1-10 copies of HIV proviral DNA per ml of blood.
It uses PCR technology to amplify proviral DNA
This test is costly and needs sophisticated instruments and highly skilled professional
It is highly sensitive and the chance of false positivity is high. hence it should not be used for making initial diagnosis of HIV infection.
Uses of DNA PCR;
i. To make early diagnosis of HIV in HIV exposed infants as serology tests are unable to diagnose HIV till the infant is 18 months old
ii. To diagnose or confirm virologic failure in patients who are not responding to ART
iii. When there is indeterminate serology
iv. For quality control of HIV rapid test.
3. CD4 T cell count :
CD4 cells play a crucial role in the body defense mechanism, measuring the amount of CD4 cells is an important indicator of the level of immune suppression that a patient infected with HIV has;
i. The average CD4 count of a normal person is in the range of 1000-1200/mm3.
ii. In patients with HIV CD4 count drops by an average of 50 - 100 cells per year
iii. Tells you the level of immune damage inflicted by HIV.
iv. It should never used to make diagnosis of HIV
v. CD4 count may be variable depending on circumstances
a. Diurnal variation; High evening low at midnight
b. Inter current infection, use of steroids and stress could affect CD4 count
vi. Following the trend in CD4 count is us full in clinical decision making.
vii. Percentage of CD4 count is useful in children below 6 years
viii. Importance’s of CD4 count
a. To decide eligibility of a patient for ART
b. To follow the progress of a patient on ART
c. To diagnose immunologic failure in patients who are not responding well to ART
Additional tests in HIV infection include:
1. HBV , HCV
2. FBP and ESR
3. AST and ALT
4. Serum Creatinine
5. Syphilis serology: RPR
6. AFB chest X-Ray where possible
7. Stool examination for parasites
8. Malaria blood slide
9. Pregnancy test for women in child bearing age
Click here to access Unit one Content..
Topic 2.6: HIV/AIDS (CDC and WHO Classification of HIV)
CDC CLASSIFICATION SYSTEM FOR HIV INFECTION
The CDC categorization of HIV/AIDS is based on the lowest documented CD4 cell count and on previously diagnosed HIV-related conditions. For example, if a patient had a condition that once met the criteria for category B but now is asymptomatic, the patient would remain in category B. Additionally, categorization is based on specific conditions, as indicated below. Patients in categories A3, B3, and C1-C3 are considered to have AIDS.
CDC Classification System for HIV-Infected Adults and Adolescents
|
CD4 Cell Categories |
Clinical Categories |
||
|
Abbreviations: PGL = persistent generalized lymphadenopathy |
|||
|
A |
B* |
C# |
|
|
(1) ≥500 cells/µL |
A1 |
B1 |
C1 |
|
(2) 200-499 cells/µL |
A2 |
B2 |
C2 |
|
(3) <200 cells/µL |
A3 |
B3 |
C3 |
* Category B Symptomatic Conditions
Category B symptomatic conditions are defined as symptomatic conditions occurring in an HIV-infected adolescent or adult that meet at least one of the following criteria:
- They are attributed to HIV infection or indicate a defect in cell-mediated immunity.
- They are considered to have a clinical course or management that is complicated by HIV infection.
Examples include, but are not limited to, the following:
- Bacillary angiomatosis
- Oropharyngeal candidiasis (thrush)
- Vulvovaginal candidiasis, persistent or resistant
- Pelvic inflammatory disease (PID)
- Cervical dysplasia (moderate or severe)/cervical carcinoma in situ
- Hairy leukoplakia, oral
- Herpes zoster (shingles), involving two or more episodes or at least one dermatome
- Idiopathic thrombocytopenic purpura
- Constitutional symptoms, such as fever (>38.5ºC) or diarrhea lasting >1 month
- Peripheral neuropathy
# Category C AIDS-Indicator Conditions
- Bacterial pneumonia, recurrent (two or more episodes in 12 months)
- Candidiasis of the bronchi, trachea, or lungs
- Candidiasis, esophageal
- Cervical carcinoma, invasive, confirmed by biopsy
- Coccidioidomycosis, disseminated or extrapulmonary
- Cryptococcosis, extrapulmonary
- Cryptosporidiosis, chronic intestinal (>1 month in duration)
- Cytomegalovirus disease (other than liver, spleen, or nodes)
- Encephalopathy, HIV-related
- Herpes simplex: chronic ulcers (>1 month in duration), or bronchitis, pneumonitis, or esophagitis
- Histoplasmosis, disseminated or extrapulmonary
- Isosporiasis, chronic intestinal (>1-month duration)
- Kaposi sarcoma
- Lymphoma, Burkitt, immunoblastic, or primary central nervous system
- Mycobacterium avium complex (MAC) or Mycobacterium kansasii, disseminated or extrapulmonary
- Mycobacterium tuberculosis, pulmonary or extrapulmonary
- Mycobacterium, other species or unidentified species, disseminated or extrapulmonary
- Pneumocystis jiroveci (formerly carinii) pneumonia (PCP)
- Progressive multifocal leukoencephalopathy (PML)
- Salmonella septicemia, recurrent (nontyphoid)
- Toxoplasmosis of brain
- Wasting syndrome caused by HIV (involuntary weight loss >10% of baseline body weight) associated with either chronic diarrhea (two or more loose stools per day for ≥1 month) or chronic weakness and documented fever for ≥1 month
WHO Clinical Staging of HIV/AIDS and Case Definition
The clinical staging and case definition of HIV for resource-constrained settings were developed by the WHO in 1990 and revised in 2007. Staging is based on clinical findings that guide the diagnosis, evaluation, and management of HIV/AIDS, and it does not require a CD4 cell count. This staging system is used in many countries to determine eligibility for antiretroviral therapy, particularly in settings in which CD4 testing is not available. Clinical stages are categorized as 1 through 4, progressing from primary HIV infection to advanced HIV/AIDS. These stages are defined by specific clinical conditions or symptoms. For the purpose of the WHO staging system, adolescents and adults are defined as individuals aged ≥15 years.
WHO CLINICAL STAGING OF HIV/AIDS FOR ADULTS AND ADOLESCENTS
|
Primary HIV Infection |
|
|
Clinical Stage 1 |
|
|
Clinical Stage 2 |
|
|
Clinical Stage 3 |
|
|
Clinical Stage 4 |
|
Click here to access Unit one Content..
Topic 2.7: HIV/AIDS (TREATMENT AND PREVENTION OF HIV)
Classes of HIV/AIDS Antiretroviral Drugs;
The antiretroviral medications used to treat HIV/AIDS currently are organized into five major drug classes.
Reverse Transcriptase (RT) Inhibitors interfere with the critical step during the HIV life cycle known as reverse transcription. During this step, the HIV enzyme RT converts HIV RNA to HIV DNA. There are two main types of RT inhibitors.
1) Nucleoside/nucleotide RT inhibitors are faulty DNA building blocks. When these faulty pieces are incorporated into the HIV DNA (during the process when HIV RNA is converted to HIV DNA), the DNA chain cannot be completed, thereby blocking HIV from replicating in a cell.
2) Non-nucleoside RT inhibitors bind to RT, interfering with its ability to convert the HIV RNA into HIV DNA.
3) Protease Inhibitors interfere with the protease enzyme that HIV uses to produce infectious viral particles.
4) Fusion/Entry Inhibitors interfere with the virus' ability to fuse with the cellular membrane, thereby blocking entry into the host cell.
5) Integrase Inhibitors block integrase, the enzyme HIV uses to integrate genetic material of the virus into its target host cell.
Multidrug Combination Products combine drugs from more than one class into a single product. To combat virus strains from becoming resistant to specific antiretroviral drugs, healthcare providers recommend that people infected with HIV take a combination of antiretroviral drugs known as highly active antiretroviral therapy (HAART). Developed by NIAID-supported researchers, the HAART strategy combines drugs from at least two different antiretroviral drug classes.
In Development
Another HIV/AIDS drug class known as maturation inhibitors is still in development. If successful, they could potentially prevent HIV from properly assembling and maturing. For example, these treatments could block HIV from forming a protective outer coat or from emerging from human cells.
Prevention of HIV
Currently, there is no vaccine to prevent HIV infection nor is there a cure for HIV/AIDS. To reduce your risk of becoming infected with HIV or transmitting the virus to others
- Get tested regularly for HIV
- Practice abstinence
- Remain faithful to your spouse or partner
- Consistently use male latex or female polyurethane condoms
- Do not share needles
Click here to access Unit Two Content..
Topic 1.1: ANTHRAX
Aetiology;
Bacillus anthracis, a spore-forming, gram-positive rod that is aerobic or facultatively anaerobic.
The name comes from the Greek word for “coal,” a reference to the black eschar that eventually forms in the cutaneous form of anthrax.
Epidemiology;
Human infection with B. anthracis is usually linked to a zoonotic source such as goats, sheep, cattle, antelope, kudu, pigs, horses, zebu, and other animals.
Animal-related products that can transmit the infection include meat, wool, hides, bones, and hair. Soil contaminated with spores that can persist for many years is also a source of infection. The spores can infect humans or animals, after which they germinate into the vegetative form of the bacteria and then cause clinical disease. Infection has been reported from exposure to cadavers from disturbed graves of individuals or animals that dies from anthrax.
Pathophysiology;
The major virulence factors of B. anthracis include its two binary toxins,
1. Edema factor(EF) and
2. Lethal factor (LF), and its antiphagocytic poly-d-glutamic acid capsule.
Both EF and LF must first bind a third anthrax toxin component, “protective antigen” (PA), to bind to a cellular “anthrax toxin receptor” (ATR).
The pathogenesis of anthrax is linked to its two binary toxins. Two cellular receptors serve as the ATR:
1) Tumor endothelial marker 8 (TEM8) and
2) Capillary morphogenesis protein 2 (CMG2).
Once the PA component of the edema toxin and lethal toxin binds to a cellular receptor, the toxin complex can enter the target cell, first going into the acidic endosome and then the cytosol.
The primary mechanism of action of lethal toxin is to inhibit the mitogen-activated protein kinase (“MAP kinase”) intracellular signal transduction pathway. Lethal toxin is linked to excessive production of inflammatory cytokines and dyregulation of the cytokine network, possibly leading to a “cytokine storm” that contributes to rapid multiorgan clinical failure, shock, and death.
Lethal toxin is also reported to impair platelets, again by inhibiting MAP kinase pathways, such as p38 MAP kinase. In addition, lethal toxin can impair human whole blood clotting in vitro. Thus, lethal toxin may contribute in several ways to the striking bleeding diathesis, including bleeding into lymph nodes, the pleural space, the meninges, the intestines, and ascitic fluid.
Clinical features;
Four major clinical manifestations of anthrax infection include;
1. Inhalational,
2. Cutaneous,
3. Gastrointestinal, and
4. Meningeal
Are related to the three routes by which B. anthracis can enter the body: inhalation, ingestion, or via the skin.
1. Inhalational anthrax;
Typically causes mediastinal adenopathy that results in mediastinal widening, as well as pleural effusions that are often bloody, can expand rapidly, recur if drained by thoracentesis rather than by chest tube, and can be rapidly fatal if not treated with multiple antibiotics and pleural drainage.
Radiography demonstrated either pulmonary infiltrates or consolidation in 8 of the 11 patients. The incubation period of inhalational anthrax is generally 3 to 14 days, with a range of 2 to 43 days in humans. In all six of the patients who survived the anthrax attacks in 2001, anthrax was diagnosed during the “intermediate-progressive stage,” and prompt therapy prevented progression of disease beyond this stage.
2. Cutaneous anthrax;
Cutaneous anthrax is the most common form of the disease (95%).
The incubation period is usually 1 to 12 days. Cutaneous anthrax typically progresses from a papule that is pruritic, to a central vesicular or bullous lesion with surrounding nonpitting edema, and finally to a necrotic and hemorrhagic central lesion that evolves into the classic painless black eschar with surrounding edema.
This sequence of progressive cutaneous lesions can still occur even if appropriate antibiotics are given, and total resolution can take up to 2 months.
Differential diagnosis of cutaneous anthrax;
i. Brown recluse spider bites (which typically cause a painful lesion unlike the painless anthrax lesion;
ii. Cutaneous leishmaniasis,
iii. Plague,
iv. Tularemia,
v. Typhus, and
vi. Warfarin (Coumadin) or
vii. Heparin necrosis.
3. Gastrointestinal anthrax;
Gastrointestinal anthrax can be divided into;
i. Oropharyngeal form and
ii. Intestinal form.
Gastrointestinal anthrax is often linked to consumption of the meat of infected animals, thereby suggesting that the vegetative form of the bacteria can cause gastrointestinal disease in humans.
The oropharyngeal form is accompanied by painful cervical adenopathy and fever. The incubation period ranges from 2 to 144 hours. Oral lesions can ulcerate and progress to cause a white pseudomembrane, hoarseness, and dysphagia.
Three phases of intestinal anthrax;
1) Prodromal phase with fever, malaise, and syncope;
2) Second progressive phase with abdominal pain, nausea, vomiting, abdominal distention, ascites, and severe weakness;
3) Fulminant third phase with suddenly increasing abdominal girth and expanding ascites, paroxysmal abdominal pain, and shock.
4. Meningeal anthrax;
Anthrax meningoencephalitis can occur in association with inhalational, cutaneous, or gastrointestinal anthrax, and in rare cases it occurs without a known portal of entry. Anthrax meningitis is nearly always fatal. Cerebral edema, parenchymal brain hemorrhages, vasculitis, and subarachnoid hemorrhages can be seen.
Diagnosis;
1. Blood should be obtained immediately for culture before any antibiotics are started; patients with inhalation anthrax will grow the typical large, gram-positive rods in less than 24 hours. Surprisingly, even one dose of an effective antibiotic can sterilize the blood cultures. Nasal cultures should not be performed as a clinical diagnostic test for individual patients but might be useful as part of an epidemiologic investigation. If the initial culture is suggestive, three additional tests that should be performed in a biosafety cabinet are motility (nonmotile), catalase (positive), and hemolysis (negative).
2. Sample is sent to a reference laboratory where polymerase chain reaction can identify B. anthracis and gamma-phage lysis of the encapsulated bacteria can provide confirmation.
3. Anthrax Quick ELISA (enzyme-linked immunosorbent assay) test can detect the eventual presence of antibodies to anthrax in blood.
4. Non–contrast-enhanced computed tomography scan can be a valuable adjunctive diagnostic tool for inhalational anthrax and is better than chest radiography for demonstrating the characteristic hyperattenuating mediastinal adenopathy and pleural effusions seen with inhalational anthrax. The hyperattenuation is consistent with bleeding into the lymph nodes and thus is helpful in distinguishing anthrax from tularemia, histoplasmosis, tuberculosis, sarcoidosis, and most other causes of hilar or mediastinal adenopathy.
Anthrax meningitis is typically bloody and neutrophilic. Large gram-positive rods distinguish it from other causes of gram-positive rod meningitis, such as the smaller Listeria monocytogenes
Treatment of Anthrax;
Treatment of clinical anthrax includes antibiotics and, in the case of inhalational anthrax, pleural fluid drainage.
The CDC recommendations for treatment include a multidrug regimen with either;
1. Ciprofloxacin (400 mg intravenously every 12 hours)
2. Doxycycline (100 mg intravenously every 12 hours) plus one or more addi-tional antimicrobials for inhalational or gastrointestinal anthrax:
3. Rifampicin,
4. Vancomycin,
5. Penicillin,
6. Ampicillin,
7. Chloramphenicol,
8. Imipenem,
9. Clindamycin, or
10. Clarithromycin.
Penicillin (or ampicillin) should not be used as a single agent in the initial therapy for inhalational anthrax because of the risk that resistance will develop.
Initial treatment of suspected anthrax meningitis should be combination therapy with a fluoroquinolone given intravenously and one or two other antibiotics that penetrate well into the central nervous system and are active against B. anthracis, such as penicillin, ampicillin, meropenem, vancomycin, or rifampicin.
Prevention of Anthrax;
1. Pre exposure vaccination for animal handlers and other high risk individuals.
2. Pre exposure antibiotics with Ciprofloxacin and Doxycycline.
3. Prevention of bioterrorism.
4. Proper disposal of carcase by cremation or deep burying.
Click here to access Unit Two Content..
Topic 1.2: BRUCELLOSIS
This is a bacterial zoonosis transmitted directly or indirectly to humans from infected animals, predominantly domesticated ruminants and swine.
The fever in brucellosis has a remittent/undulating character.
Humans are affected by contact with affected animals and infectious animal products.
Aetiology;
Brucella species;
Variants include;
1. B. melitensis, which is the most common cause of symptomatic disease in humans and for which the main sources are sheep, goats, and camels;
2. B. abortus, which is usually acquired from cattle or buffalo:
3. B. suis, which generally is acquired from swine but has one variant enzootic in reindeer and caribou and another in rodents;
4. B. canis, which is acquired most often from dogs.
5. B. ovis, which causes reproductive disease in sheep,
6. B. neotomae, which is specific for desert rodents, have not been clearly implicated in human disease.
All brucellae are small, gram-negative, unencapsulated, nonsporulating, nonmotile rods or coccobacilli. They are facultative aerobes.
They grow aerobically on peptone-based medium incubated at 37°C; the growth of some types is improved by supplementary CO2. In vivo, brucellae behave as facultative intracellular parasites.
The organisms are sensitive to sunlight, ionizing radiation, and moderate heat; they are killed by boiling and pasteurization but are resistant to freezing and drying.
Their resistance to drying renders brucellae stable in aerosol form, facilitating airborne transmission. This ability enable the organism to have a potential for bioterrorism and therefore able to be weaponized. The inoculum required for an infection is as low as 10-100 microorganisms.
Epidemiology
Brucellosis is a zoonosis whose occurrence is closely related to its prevalence in domesticated animals.
The true global prevalence of human brucellosis is unknown because of the imprecision of diagnosis and the inadequacy of reporting and surveillance systems in many countries.
There are differences in virulence among the four species;
1) B. abortus, with a reservoir in cattle, is usually associated with mild to moderate sporadic disease; suppurative or disabling complications are rare.
2) B. suis infection, resulting from swine contact, is often associated with destructive, suppurative lesions and may have a prolonged course.
3) B. melitensis, with a reservoir in sheep and goats, may cause severe, acute disease and disabling complications.
4) B. canis, spread to humans from infected dogs, causes disease with an insidious onset, frequent relapse, and a chronic course that is indistinguishable from that of infection related to B. abortus.
Studies indicate that there are two new species, Brucella pinnipediae and Brucella cetaceae, related to seals and cetaceans, respectively. Two cases of human marine-associated Brucella spp infection have been reported.
Human brucellosis;
This is usually associated with occupational or domestic exposure to infected animals or their products.
Farmers, shepherds, goatherds, veterinarians, and employees in slaughterhouses and meat-processing plants in endemic areas are occupationally exposed to infection.
Family members of individuals involved in animal husbandry may be at risk, although it is often difficult to differentiate food-borne infection from environmental contamination under these circumstances.
Laboratory workers who handle cultures or infected samples are also at risk. Travelers and urban residents usually acquire the infection through consumption of contaminated foods. Person-to-person transmission is extremely rare, as is transfer of infection by blood or tissue donation. Although brucellosis is a chronic intracellular infection, there is no evidence for increased prevalence or severity among individuals with HIV infection or with immunodeficiency or immunosuppression of other etiologies.
Portal of entry of the infection;
Brucellosis may be acquired by;
1. Ingestion,
2. Inhalation, or
3. Mucosal or percutaneous exposure.
B. melitensis and B. suis have been developed as biological weapons by several countries and could be exploited for bioterrorism. This possibility should be borne in mind in the event of sudden unexplained outbreaks.
The organism can also be transmitted sexually.
Immunity and Pathogenesis
Exposure to brucellosis elicits both humoral and cell-mediated immune responses.
Antibodies promote clearance of extracellular brucellae by bactericidal action and by facilitation of phagocytosis by polymorphonuclear and mononuclear phagocytes; however, antibodies alone cannot eradicate infection.
Tumor necrosis factor (TNF-) produced early in the course of infection stimulates cytotoxic lymphocytes and activates macrophages, which can kill intracellular brucellae (probably mainly through production of reactive oxygen and nitrogen intermediates) and may clear infection. However, virulent Brucella cells can suppress the TNF- response, and control of infection in this situation depends on macrophage activation and interferon (IFN-) responses.
Cytokines such as interleukin (IL) 12 promote production of IFN-, which drives TH1-type responses and stimulates macrophage activation. Inflammatory cytokines, including IL-4, IL-6, and IL-10, downregulate the protective response.
After penetrating the epithelial cells of human skin, conjunctiva, pharynx, or lung, Brucella organisms initially induce an exuberant polymorphonuclear neutrophil response in the submucosa. After ingestion of organisms by neutrophils and tissue macrophages, spread to regional lymph nodes occurs. If host defenses within the lymph nodes are overwhelmed, bacteremia follows.
Clinical Features;
1. Fever is the most common symptom and sign of brucellosis, occurring in 80-100% of cases.
1) It is intermittent in 60% of patients with acute and chronic brucellosis and undulant in 60% of patients with subacute brucellosis.
2) Fever can be associated with a relative bradycardia.
3) Fever of unknown origin (FUO) is a common initial diagnosis in patients in areas of low endemicity.
4) It is associated with chills in almost 80% of cases.
2. Constitutional symptoms of brucellosis include;
1) Anorexia,
2) Asthenia,
3) Fatigue,
4) Weakness, and malaise, and
5) weight loss and are very common (> 90% of cases).
3. Bone and joint symptoms include
1) Arthralgias,
2) Low back pain,
3) Spine and joint pain, and, rarely, joint swelling. These symptoms affect as many as 55-80% of patients. Arthralgias may be diffuse or localized, with a predilection for bone ends and the sacroiliac joint. Acute monoarticular arthritis is uncommon but may be part of the presentation.
4. Neuropsychiatric symptoms of brucellosis are common despite the rare involvement of the nervous system.
1) Headache,
2) Depression, and
3) Fatigue are the most frequently reported neuropsychiatric symptoms. In patients with advanced disease who have meningoencephalitis, these complaints may include changes in mental status, coma, neurologic deficit, nuchal rigidity, or seizures.
5. Neurologic symptoms of brucellosis can include weakness, dizziness, unsteadiness of gait, and urinary retention. Symptoms associated with cranial nerve dysfunction may affect persons with chronic central nervous system (CNS) involvement.
6. A significant percentage (approximately 50%) of patients have gastrointestinal (GI) complaints, primarily dyspepsia, though abdominal pain from hepatic abscesses may occur. Hepatic abscesses should be suspected in patients with signs of systemic toxicity and persistently elevated liver enzymes. The abscess can serve as a source of bacteremic seeding. Spontaneous bacterial peritonitis secondary to brucellosis infection has been reported. Constipation, diarrhea, and vomiting may occur.
7. Genitourinary infections with brucellae have been reported and include orchitis, urinary tract infection (UTI), and glomerulonephritis. Frank renal failure or sepsis is rare.
8. Cough and dyspnea develop in up to 19% of persons with brucellosis; however, these symptoms are rarely associated with active pulmonary involvement. Pleuritic chest pain may affect patients with underlying empyema.
9. Endocarditis from brucellae is reported, with septic embolization a common complication of this form of brucellosis. Other cardiac complications, such as pulmonary edema or dysrhythmias, are rare. Brucella endocarditis is the form most commonly associated with fatalities.
Chronic brucellosis;
With the chronic form of brucellosis, in which the illness has lasted longer than 1 year (undiagnosed and untreated brucellosis), an afebrile pattern is typical, with a history of myalgia, fatigue, depression, and arthralgias (chronic fatigue syndrome is the most important disease in the differential diagnosis).
The chronic form is primarily caused by B melitensis and usually affects adults older than 30 years. The chronic form is rare in children.
Physical Examination findings in brucellosis;
Generally, physical examination findings are normal or only minimally abnormal and the diagnosis is made on the basis of the history and serologic studies.
Categorization of Brucellosis;
1) Subclinical,
2) Acute,
3) Subacute, or
4) chronic;
5) Localized and relapsing forms have also been described.
This classification system, though commonly used, is subjective and of limited clinical utility.
1) Subclinical brucellosis/Asymptomatic
Disease is usually asymptomatic, and the diagnosis is usually established incidentally after serologic screening of persons at high risk of exposure. Culture data are usually unrevealing.
2) Acute and subacute brucellosis
Disease can be mild and self-limited (eg, B abortus) or fulminant with severe complications (eg, B melitensis). Associated symptoms can develop 2-3 months before diagnosis in mild cases and 3-12 months before diagnosis in severe cases.
Usually, acute brucellosis occurs without focal abnormalities. Nonfocal weakness may be noted. The tissues overlying the spine or peripheral nerves may be tender to percussion. Tenderness, swelling, or effusion of joints may be evident. In some instances, orchitis appears after a few days of illness. Testicular swelling and tenderness in the wake of chills and high fever thus resemble mumps orchitis.
Some patients manifest constipation. Occasionally, abdominal tenderness suggests an acute abdomen. In some more severe cases, tender enlargement of the spleen may be detected.
3) Chronic brucellosis
The diagnosis of chronic brucellosis is typically made after symptoms have persisted for 1 year or more. Low-grade fevers and neuropsychiatric symptoms predominate. Results of serologic studies and cultures are often negative; without confirmatory evidence, many authorities doubt the existence of chronic disease. Many patients have persistent disease caused by inadequate initial therapy, and underlying localized disease may be present.
4) Localized and relapsing brucellosis
Localized complications of brucellosis are typically observed in patients with acute disease or chronic untreated infection. Osteoarticular, genitourinary, and hepatosplenic involvement are most common. Cultures of involved tissue sites and serology can be diagnostic.
Relapsing brucellosis may be difficult to distinguish from reinfection. Presenting symptoms typically reflect the initial disease; however, these symptoms are more severe. Symptoms typically develop 2-3 months after therapy completion. Culture results are typically positive, and serology may be difficult to interpret, but enzyme-linked immunoassay (ELISA) testing may be more help
Complications of brucellosis;
Complications are rare in the patient who is treated appropriately, though relapse of infection may occur in 10% of patients. The major risk factor for the development of focal complications is symptom duration greater than 30 days before diagnosis. Others include inadequate medication or poor adherence to treatment.
1. Splenic abscess
2. Thyroid abscess
3. Epidural abscess
4. Uveitis
5. Neurobrucellosis.
6. Abortion.
Differential Diagnoses of Brucellosis;
1. Febrile illnesses;
1) Malaria.
2) Enteric fever.
2. Migrain Headache.
3. Hypertension.
4. Early pregnancy syndrome.
5. HIV and other pyrexia of unknown origin.
6. Tuberculosis.
7. Abortion Complications
8. Acute Epididymitis
9. Ankylosing Spondylitis and Undifferentiated Spondyloarthropathy
10. Bacterial Pneumonia
11. Brain Abscess in Emergency Medicine
12. Bronchitis
Laboratory Investigation;
1. Baseline Tests;
1) Complete blood count
A complete blood count (CBC) typically is ordered routinely as part of an evaluation for a patient with potential infectious disease. Leukocytosis is rare in brucellosis, and a significant number of patients are neutropenic.
Anemia is reported in 75% of patients (particularly with chronic infection), thrombocytopenia is reported in 40% (secondary to hepatosplenomegaly or from immune thrombocytopenia), and pancytopenia is reported in 6% of patients.
2) Liver enzymes
A slight elevation in liver enzyme levels is a very common finding. These elevated levels may reflect the severity of hepatic involvement and correlate clinically with hepatomegaly.
2. Specific tests;
1) Serology/BAT- Brucella Antigen Test.
Serologic testing is the most commonly used method of diagnosing brucellosis. Repeated serologic testing is recommended if the initial titer is low.
The tube agglutination test, developed by Bruce, measures antibodies against smooth lipopolysaccharide (LPS); it remains the most popular test tool for the diagnosis of brucellosis. The 2-mercaptoethanol test detects immunoglobulin G (IgG), and titers higher than 1:80 define active infection. A high IgG antibody titer or a titer that is higher after treatment suggests persistent infection or relapse. Other tests, such as tray agglutination (TAT) and modified TAT, are also popular.
Titers higher than 1:160 in conjunction with a compatible clinical presentation are considered highly suggestive of infection. Titers higher than 1:320 are considered to be more specific, especially in endemic areas. Seroconversion and evolution of the titers can also be used for diagnosis.
The shortcomings of agglutination tests test include potential cross-reactivity with IgM of other organisms such as Francisella tularensis, Salmonella urbana, Yersinia enterocolitica serotype O9, Vibrio cholerae, Afipia clevelandensis, and some other bacteria.
Prozone phenomenon may occur secondary to hyperantigenemia, possibly leading to false-negative results. Accordingly, routine dilution of sera (typically beyond 1:320) is necessary to avoid this problem.
2) Culture
Diagnosis of brucellosis is definitive when Brucella organisms are recovered from blood, bone marrow, or other tissue. Some Brucella species require 5-10% carbon dioxide for primary isolation. Because of the ease of aerosol transmission, any potential Brucella specimens should be handled under a biohazard hood.
The sensitivity of blood cultures with improved techniques such as the Castaneda bottles is further improved by the lysis-centrifugation technique. With these methods, the sensitivity is approximately 60%.
Subcultures are still advised for at least 4 weeks; thus, if brucellosis is suspected, the laboratory should be alerted to keep the cultures for 3-4 weeks, which is not done routinely for most bacterial cultures.
Because the reticuloendothelial system holds a high concentration of brucellae, bone marrow culture is thought to be the criterion standard. Sensitivity is usually 80-90%.
NOTE; Any fluid (eg, synovial fluid, pleural fluid, or cerebrospinal fluid [CSF]) can be cultured, but the yield is usually low.
3) CSF analysis
Mostly in patients with neurobrucellosis, analysis of CSF reveals a mild-to-modest lymphocytic pleocytosis of 88-98%. Protein levels are elevated in conjunction with normal glucose levels. CSF cultures are positive for brucellosis less than 50% of the time, but antibody testing of the fluid yields a diagnosis. CSF cultures are indicated for suggested meningitis.
4) Arthrocentesis
Although significant joint effusion is uncommon, arthrocentesis may occasionally be needed to exclude septic arthritis. The joint aspirate demonstrates an exudative fluid with low cell counts and mononuclear predominance. Patients with brucellosis rarely present with acute monoarticular arthritis.
Rx of Brucellosis;
The recommended minimum duration of treatment is 6 weeks.
1. Treatment In Adults.
2. Treatment in Pregnant/expecting women.
3. Treatment in Children
4. Treatment of Neurobrucellosis.
5. Treatment of Chronic Brucellosis.
The World Health Organization (WHO) guidelines recommend the following:
1) Treatment In adults and Children >8 years.
i. Option 1;
Doxycycline 100 mg PO twice daily plus rifampin 600-900 mg/day PO – Both drugs are to be given for 6 weeks; this regimen is more convenient but probably increases the risk of relapse.
A 2012 Cochrane review found that a regimen consisting of doxycycline for 6 weeks plus streptomycin for 2-3 weeks was more effective than one consisting of doxycycline plus rifampicin for 6 weeks. The investigators also found that a regimen consisting of a fluoroquinolone plus rifampicin for 6 weeks was as effective overall as doxycycline plus rifampin (though the evidence for this conclusion was of low quality) and was slightly better tolerated.
ii. Option 2;
Doxycycline 100 mg PO twice daily for 6 weeks and streptomycin 1 g/day IM for 2-3 weeks – This regimen is believed to be more effective, mainly in preventing relapse; gentamicin can be used as a substitute for streptomycin and has shown equal efficacy. Gentamicin is given Once a day for at least 7 days.
iii. Option 3:
Ciprofloxacin-based regimens have shown efficacy equal to that of doxycycline-based regimens. However Doxycycline based combination is preffered.
2) Children Younger than 8 years;
For brucellosis in children younger than 8 years, administration of rifampin and TMP-SMZ for 6 weeks is the therapy of choice. The relapse rate appears to be approximately 5% or lower.
3) Treatment of brucellosis in pregnant women;
This is a challenging problem, and the available data are limited. TMP-SMZ has been effective in this population, either as monotherapy or as part of combination therapy with rifampin or gentamicin. The most common recommendation is for rifampin, either alone or in combination with TMP-SMZ. It should be kept in mind that the use of TMP-SMZ by the end of pregnancy is associated with kernicterus.
In patients with spondylitis or sacroiliitis, doxycycline and rifampin combined with an aminoglycoside (gentamicin) for the initial 2-3 weeks, followed by 6 weeks of rifampin and doxycycline, is usually recommended.
4) Treatment of Neurobrucellosis;
Patients with nervous system infections typically require combination therapy/Tripple therapy.
1. Doxycycline is generally preferred to tetracyclines or aminoglycosides because the latter are less likely to cross the blood-brain barrier in adequate quantities.
2. Many authorities prefer a 3-drug (doxycycline-streptomycin-rifampin or doxycycline−TMP-SMZ−rifampin) regimen to a 2-drug regimen. A brief course of adjunctive corticosteroid therapy has been used to control the inflammatory process.
5) Chronic brucellosis;
This is treated with triple-antibiotic therapy. The combination of rifampin, doxycycline, and streptomycin often is used.
Corticosteroids and other agents;
The use of corticosteroids is reserved for symptomatic Brucella meningitis. Although these agents are generally recommended, scientific evidence supporting their use is lacking. No consensus exists on optimal dosing, frequency, or duration of therapy.
Any additional drugs needed for symptomatic treatment (eg, antipyretics, analgesics) must be administered as well. Additional medication is based on the patient’s presenting symptoms.
Prevention Of Brucellosis;
1. Vaccines based on live attenuated Brucella strains, such as B. abortus strain 19BA or 104M, have been used in some countries to protect high-risk populations but have displayed only short-term efficacy and high reactogenicity. Subunit vaccines have been developed but are of uncertain value and cannot be recommended at present. Research in this area has been stimulated by interest in biodefense and may eventually yield new products, some of which may be based on the live attenuated WR 201 variant of B. melitensis strain 16M.
2. The mainstay of veterinary prevention is a national commitment to testing and slaughter/culling of infected herds/flocks (with compensation for owners), control of animal movement, and active immunization of animals. These measures are usually sufficient to control human disease as well. In their absence, pasteurization of all milk products before consumption is sufficient to prevent non-occupational animal-to-human transmission. All cases of brucellosis in animals and humans should be reported to the appropriate public health authorities.
Click here to access Unit Two Content..
Topic 1.3: CHOLERA
Aetiology;
Vibrio cholerae.
V cholerae is a saltwater organism, and its primary habitat is the marine ecosystem where it lives in association with plankton.
Cholera has 2 main reservoirs, humans and water. V cholerae is rarely isolated from animals, and animals do not play a role in transmission of disease.
The hallmark of the disease is profuse secretory diarrhea which manifests as;
· Effortless, painless, watery diarrhea usually voluminous.
Mode of Transmission;
Cholera is transmitted by the fecal-oral route by ingestion of contaminated food, water or contact with formites.,
NOTE;
Definitive diagnosis is not a prerequisite for the treatment of patients with cholera. The priority in management of any watery diarrhea is replacing the lost fluid and electrolytes and providing an antimicrobial agent when indicated.
A new strain of cholera, V cholerae serogroup O139 (Bengal) emerged in the fall of 1992 and caused outbreaks in Bangladesh and India in 1993. Disease from this strain has become endemic in at least 11 countries.
Cholera has been rare in industrialized nations for the past century; however, the disease is still common in other parts of the world, including the Indian subcontinent and sub-Saharan Africa. Epidemics occur after war, civil unrest, or natural disasters when water and food supplies become contaminated with V cholerae in areas with crowded living conditions and poor sanitation.
Pathophysiology;
The pathogen;
V cholerae is a comma-shaped, gram-negative aerobic or facultatively anaerobic bacillus that varies in size from 1-3 µm in length by 0.5-0.8 µm in diameter (see the image below).
Its antigenic structure consists of a flagellar H antigen and a somatic O antigen. The differentiation of the latter allows for separation into pathogenic and nonpathogenic strains. Although more than 200 serogroups of V cholerae have been identified, V cholerae O1 and V cholerae O139 are the principal ones associated with epidemic cholera.
Currently, the El Tor biotype of V cholerae O1 is the predominant cholera pathogen.
Organisms in both the classical and the El Tor biotypes are subdivided into serotypes according to the structure of the O antigen, as follows:
1. Serotype Inaba - O antigens A and C
2. Serotype Ogawa - O antigens A and B
3. Serotype Hikojima - O antigens A, B, and C
The clinical and epidemiologic features of disease caused by V cholerae O139 are indistinguishable from those of disease caused by O1 strains. Both serogroups cause clinical disease by producing an enterotoxin that promotes the secretion of fluid and electrolytes into the lumen of the small intestine.
Pathology;
To reach the small intestine, however, the organism has to negotiate the normal defense mechanisms of the GI tract. Because the organism is not acid-resistant, it depends on its large inoculum size to withstand gastric acidity.
The infectious dose of V cholerae required to cause clinical disease varies by the mode of administration. If V cholerae is ingested with water, the infectious dose is 103 -106 organisms. When ingested with food, fewer organisms (102 -104) are required to produce disease.
The use of antacids, histamine receptor blockers, and proton pump inhibitors increases the risk of cholera infection and predisposes patients to more severe disease as a result of reduced gastric acidity. The same applies to patients with chronic gastritis secondary to Helicobacter pylori infection or those who have undergone a gastrectomy.
V cholerae O1 and V cholerae O139 cause clinical disease by producing an enterotoxin that promotes the secretion of fluid and electrolytes into the lumen of the small intestine. The enterotoxin is a protein molecule composed of 5 B subunits and 2 A subunits. The B subunits are responsible for binding to a ganglioside (monosialosyl ganglioside, GM1) receptor located on the surface of the cells that line the intestinal mucosa.
Cause of secretory diarrhea;
The A1 subunit of cholera toxin activates adenylate cyclase to cause a net increase in cyclic adenosine monophosphate (cAMP). The increased cAMP then carries on the downstream effects. cAMP blocks the absorption of sodium and chloride by the microvilli and promotes the secretion of chloride and water by the crypt cells. The result is watery diarrhea with electrolyte concentrations isotonic to those of plasma.
Fluid loss originates in the duodenum and upper jejunum; the ileum is less affected. The colon is usually in a state of absorption because it is relatively insensitive to the toxin. However, the large volume of fluid produced in the upper intestine overwhelms the absorptive capacity of the lower bowel, resulting in severe diarrhea. Unless the lost fluid and electrolytes are replaced adequately, the infected person may develop shock from profound dehydration and acidosis from loss of bicarbonate.
The enterotoxin acts locally and does not invade the intestinal wall. As a result, few neutrophils are found in the stool.
History of Presenting illness;
After a 24- to 48-hour incubation period, symptoms begin with;
1. The sudden onset of painless, effortless watery diarrhea that may quickly become voluminous and is often followed by
2. Vomiting.
3. Accompanying abdominal cramps, probably from distention of loops of small bowel as a result of the large volume of intestinal secretions.
Fever is typically absent.
An estimated 5% of infected patients will develop cholera gravis, ie, severe watery diarrhea, vomiting, and dehydration with hypovolaemic shock.
Diarrhea;
Profuse watery diarrhea is a hallmark of cholera. Cholera should be suspected when a patient older than 5 years develops severe dehydration from acute, severe, watery diarrhea (usually without vomiting) or in any patient older than 2 years who has acute watery diarrhea and is in an area where an outbreak of cholera has occurred.
Stool volume during cholera is more than that of any other infectious diarrhea. Patients with severe disease may have a stool volume of more than 250 mL/kg body weight in a 24-hour period. Because of the large volume of diarrhea, patients with cholera have frequent and often uncontrolled bowel movements.
The stool may contain fecal material early in the course of clinical illness. The characteristic cholera stool is an opaque white liquid that is not malodorous and often is described as having a “rice water” appearance (ie, in color and consistency, it resembles water that has been used to wash or cook rice).
Vomiting;
Vomiting, although a prominent manifestation, may not always be present. Early in the course of the disease, vomiting is caused by decreased gastric and intestinal motility; later in the course of the disease it is more likely to result from acidemia.
Dehydration;
If untreated, the diarrhea and vomiting lead to isotonic dehydration, which can lead to acute tubular necrosis and renal failure.
In patients with severe disease, vascular collapse, shock, and death may ensue. Dehydration can develop with remarkable rapidity, within hours after the onset of symptoms. This contrasts with disease produced by infection from any other enteropathogen. Because the dehydration is isotonic, water loss is proportional between 3 body compartments, intracellular, intravascular, and interstitial
Physical Examination in Cholera;
Clinical signs of cholera parallel the level of volume contraction. The amount of fluid loss and the corresponding clinical signs of cholera are as follows:
1) 3-5% loss of normal body weight - Excessive thirst
2) 5-8% loss of normal body weight - Postural hypotension, tachycardia, weakness, fatigue, dry mucous membranes or dry mouth.
3) >10% loss of normal body weight - Oliguria; glassy or sunken eyes; sunken fontanelles in infants; weak, thready, or absent pulse; wrinkled "washerwoman" skin; somnolence; coma
Assessment for Dehydration in Cholera;
The World Health Organization has classified dehydration in patients with diarrhea into the following 3 categories, to facilitate treatment
1. Severe
2. Some (previously termed moderate, in the WHO criteria)
3. None (previously termed mild, in the WHO criteria)
Children without clinically significant dehydration (< 5% loss of body weight) may have increased thirst without other signs of dehydration. In children with some (ie, moderate) dehydration, cardiac output and vascular resistance are normal, and changes in interstitial and intracellular volume are the primary manifestations of illness. Skin turgor is decreased, as manifested by prolonged skin tenting in response to a skin pinch (the most reliable sign of isotonic dehydration), and a normal pulse.
For the skin pinch, it is important to pinch longitudinally (Skin contours) rather than horizontally and to maintain the pinch for a few seconds before releasing the skin. The skin pinch may be less useful in patients with marasmus (severe wasting), kwashiorkor (severe malnutrition with edema), or obesity.
In adults and children older than 5 years, other signs of severe dehydration include tachycardia, absent or barely palpable peripheral pulses, and hypotension.
Tachypnea and hypercapnia also are part of the clinical picture and are attributable to the metabolic acidosis that invariably is present in patients with cholera who are dehydrated.
Atypical presentation in Pediatric patients;
In pediatric patients, the primary signs are similar to those in adults. However, children with severe cholera may present with signs that are rarely seen in adults. A child with cholera is usually very drowsy, and coma is not uncommon. In addition, pediatric patients may have convulsions that appear to be related, in part, to hypoglycemia because patients exhibit some response to intravenous dextrose. Another significant difference from the adult presentation is that children are often febrile.
CHOLERA SICCA
Cholera sicca is an old term describing a rare, severe form of cholera that occurs in epidemic cholera. This form of cholera manifests as ileus/Paralytic ileus and abdominal distention from massive outpouring of fluid and electrolytes into dilated intestinal loops.
Mortality is high, with death resulting from toxemia before the onset of diarrhea and vomiting. The mortality in this condition is high. Because of the unusual presentation, failure to recognize the condition as a form of cholera is common.
Differential Diagnoses of Cholera;
1. Escherichia coli (E coli) Infections
2. Pediatric Gastroenteritis
3. Rotavirus
4. Food poisoning/contamination.
Diagnosis of Cholera;
Definitive diagnosis is not a prerequisite for the treatment of patients with cholera. The priority in management of any watery diarrhea is replacing the lost fluid and electrolytes and providing an antimicrobial agent when indicated. Usually Doxycycline.
Case definition of Cholera;
According to World Health Organization (WHO) standard case definition, a case of cholera is suspected when the following conditions are met:
1) In an area where the disease is not known to be present, a patient aged 5 years or older develops severe dehydration or dies from acute watery diarrhea
2) In an area with a noted cholera epidemic, a patient aged 5 years or older develops acute watery diarrhea, with or without vomiting
In endemic areas, biochemical confirmation and characterization of the isolate are usually unnecessary. However, these tasks may be worthwhile in areas where Vibrio cholerae is an uncommon isolate.
1. Stool analysis;/Also rectal swab is used in epidemics.
1) Direct microscopy/ Dark field microscopy;
If identification of the organism is required, direct microscopic examination of stool (including dark-field examination- dark field microscopy) is indicated, along with Gram stain, culture, and serotype and biotype identification.
Polymerase chain reaction (PCR) tests for identifying V cholerae have been developed. These have a high degree of sensitivity and specificity. At present, however, such tests are used for screening of food samples.
Vibrio cholerae is a gram-negative curved bacillus that is motile by means of a single flagellum. Laboratory diagnosis is required not only for identification but also for epidemiological purposes.
Although observed as a gram-negative organism, the characteristic motility of Vibrio species cannot be identified on a Gram stain, but it is easily seen on direct dark-field examination of the stool.
2) Stool Culture;
V cholerae is not fastidious in nutritional requirements for growth. However, it does need an adequate buffering system if fermentable carbohydrate is present because viability is severely compromised if the pH is less than 6, often resulting in autosterilization of the culture. Many of the selective media used to differentiate enteric pathogens do not support the growth of V cholerae.
a. Routine differential media
Colonies are lactose-negative, like all other intestinal pathogens, but sucrose-positive. When plated onto triple-sugar iron agar to screen for Salmonella and Shigella species, the organism gives the nonpathogenic pattern of an acid (yellow) slant and acid butt because of fermentation of the sucrose contained in triple-sugar iron agar.
Unlike other Enterobacteriaceae, V cholerae is oxidase-positive; hence, in countries where selective media are not available and cholera is not endemic, V cholerae should be suspected if any motile, oxidase-positive, gram-negative rod isolated on routine differential media from the stool of a patient with diarrhea produces an acid reaction on triple sugar iron agar.
b. Alkaline enrichment media
As Vibrio has the ability to grow at a high pH or in bile salts, which inhibit many other Enterobacteriaceae, peptone water (pH 8.5-9) or selective media containing bile salts (eg, thiosulfate-citrate-bile-sucrose-agar [pH 8.6]) are recommended to facilitate isolation and lab diagnosis. On thiosulfate-citrate-bile-sucrose-agar, the sucrose-fermenting V cholera grow as large, smooth, round yellow colonies that stand out against the blue-green agar.
3) Serotyping and Biotyping
Specific antisera can be used in immobilization tests. A positive immobilization test result (ie, cessation of motility of the organism) is produced only if the antiserum is specific for the Vibrio type present; the second antiserum serves as a negative control. Vibrio antisera may be unavailable in countries where cholera is not endemic. In endemic regions, this is an excellent quick method of identification, even in small laboratories.
Classic and El Tor biotypes also can be identified using the same method. This is useful for epidemiologic studies.
4) Hematologic Tests;
The major hematologic derangements in patients with cholera derive from the alterations in intravascular volume and electrolyte concentrations.
i. Acute phase reactants;
Hematocrit, serum-specific gravity, and serum protein are elevated in dehydrated patients because of resulting hemoconcentration. When patients are first observed, they generally have a leukocytosis without a left shift. There is overally elevated acute phase reactants.
ii. Metabolic Panel
a. Serum sodium is usually 130-135 mmol/L, reflecting the substantial loss of sodium in the stool.
b. Serum potassium usually is normal in the acute phase of the illness, reflecting the exchange of intracellular potassium for extracellular hydrogen ion in an effort to correct the acidosis.
c. Hyperglycemia may be present, secondary to systemic release of epinephrine, glucagon, and cortisol due to hypovolemia.
d. Patients have elevated blood urea nitrogen and creatinine levels consistent with prerenal azotemia. The extent of elevation depends on the degree and duration of dehydration.
e. A reduced bicarbonate level (< 15 mmol/L) and an elevated anion gap occur as a result of increases in serum lactate, protein, and phosphate levels. The arterial pH is usually low (approximately 7.2). Calcium and magnesium levels are usually high as a result of hemoconcentration.
Management of Cholera;
Rehydration is the first priority in the treatment of cholera. Rehydration is accomplished in 2 phases: rehydration and maintenance.
The goal of the rehydration phase is to restore normal hydration status, which should take no more than 4 hours. Set the rate of intravenous infusion in severely dehydrated patients at 50-100 mL/kg/hr.
Lactated Ringer solution is preferred over isotonic sodium chloride solution because saline does not correct metabolic acidosis.
The goal of the maintenance phase is to maintain normal hydration status by replacing ongoing losses. The oral route is preferred, and the use of oral rehydration solution (ORS) at a rate of 500-1000 mL/hr is recommended.
WHO Treatment Guidelines for Cholera;
The World Health Organization (WHO) guidelines for the management of cholera are practical, easily understood, and readily applied in clinical practice. These guidelines can be used for the treatment of any patient with diarrhea and dehydration. Diagnosis of cholera is not required to initiate hydration therapy.
1. Rehydration
The WHO has provided recommendations for fluid replacement in patients with dehydration. The recommendations include recommendations for fluid replacement for severe hydration, some dehydration, and no dehydration.
1) SEVERE DEHYDRATION
Administer intravenous (IV) fluid immediately to replace fluid deficit. Use lactated Ringer solution or, if that is not available, isotonic sodium chloride solution. If the patient can drink, begin giving oral rehydration salt solution (ORS) by mouth while the drip is being set up; ORS can provide the potassium, bicarbonate, and glucose that saline solution lacks.
NOTE; Coconut fluid has been used in epidemics especially is shortage of IV fluid.
· For patients older than 1 year, give 100 mL/kg IV in 3 hours—30 mL/kg as rapidly as possible (within 30 min) then 70 mL/kg in the next 2 hours.
· For patients younger than 1 year, administer 100 mL/kg IV in 6 hours—30 mL/kg in the first hour then 70 mL/kg in the next 5 hours.
Monitor the patient frequently. After the initial 30 mL/kg has been administered, the radial pulse should be strong and blood pressure should be normal. If the pulse is not yet strong, continue to give IV fluid rapidly. Administer ORS solution (about 5 mL/kg/h) as soon as the patient can drink, in addition to IV fluid.
Reassess the hydration status after 3 hours (infants after 6 h). In the rare case that the patient still exhibits signs of severe dehydration, repeat the IV therapy already given. If signs of some dehydration are present, continue as indicated below for some dehydration. If no signs of dehydration exist, maintain hydration by replacing ongoing fluid losses.
Routes for parenteral rehydration in cholera;
Accessing a peripheral vein is relatively easy, despite the severe dehydration. If a peripheral vein is not readily accessible, scalp veins have been used for initial rehydration. As the vascular volume is reestablished, a larger needle or catheter can be introduced in a peripheral vein.
Intraosseous routes have been used successfully in young children when veins cannot be accessed. The intraperitoneal route has been tried, but is not recommended.
ORS solution can be administered via nasogastric tube if the patient has some signs of dehydration and cannot drink or if the patient has severe dehydration and IV therapy is not possible at the treatment facility.
Overhydration;
A risk of overhydration exists with intravenous fluids; it usually first manifests as puffiness around the eyes. Continued excessive administration of intravenous fluids can lead to pulmonary edema and has been observed even in children with normal cardiovascular reserve.
Thus, it is important to monitor patients who are receiving intravenous rehydration hourly. Serum-specific gravity is an additional measure of the adequacy of rehydration.
2) SOME DEHYDRATION.
Administer ORS solution . WHO ORS contains the following
Sodium – 75 mmol/L
Chloride – 65 mmol/L
Potassium – 20 mmol/L
Bicarbonate – 30 mmol/L
Glucose – 111 mmol/L
A Satchet for reconstituting 500ml solution contains the following;
i. Glucose anhydrous BP- 6.7 g.
ii. Sodium Chloride BP- 1.3 g.
iii. Sodium Citrate BP- 1.45 g.
iv. Pottasium Chloride BP- 0.75g.
A homemade equivalent is 6 teaspoons of sugar and one half teaspoon of salt in a liter of water; a half cup of orange juice or some mashed banana can provide potassium.
Use the patient's age only when weight is unknown. The approximate amount of ORS required (in mL) also can be calculated by multiplying the patient's weight (in kg) times 75.
If the patient passes watery stools or wants more ORS solution than shown, give more. Monitor the patient frequently to ensure that the ORS solution is taken satisfactorily and to identify patients with profuse ongoing diarrhea who require closer monitoring.
Reassess the patient after 4 hours. In the rare case where signs of severe dehydration have appeared, rehydrate for severe dehydration, as above. If some dehydration is still present, repeat the procedures for some dehydration and start to offer food and other fluids. If no signs of dehydration are present, maintain hydration by replacing ongoing fluid losses.
Most patients absorb enough ORS solution to achieve rehydration, even when they are vomiting. Vomiting usually subsides within 2-3 hours, as rehydration is achieved.
Urine output decreases as dehydration develops and may cease. It usually resumes within 6-8 hours after starting rehydration. Regular urinary output (i e, every 3-4 h) is a good sign that enough fluid is being given.
3) NO SIGNS OF DEHYDRATION
Patients who have no signs of dehydration when first observed can be treated at home. Give these patients ORS packets to take home, enough for 2 days. Demonstrate how to prepare and give the solution.
Instruct the patient or the caretaker to return if any of the following signs develop:
1. Increased number of watery stools
2. Eating or drinking poorly
3. Marked thirst
4. Repeated vomiting
5. Any signs indicating other problems (eg, fever, blood in stool)
Maintenance of Hydration;
Maintain hydration of patients presenting with severe or some dehydration. Replace ongoing fluid losses until diarrhea stops.
When a patient who has been rehydrated with IV fluid or ORS solution is reassessed and has no signs of dehydration, continue to administer ORS solution to maintain normal hydration. The aim is to replace stool losses as they occur with an equivalent amount of ORS solution.
The amount of ORS solution required to maintain hydration varies greatly among patients, depending on the volume of stool passed. It is highest in the first 24 hours of treatment and is especially large in patients who present with severe dehydration. In the first 24 hours, the average requirement of ORS solution in such patients is 200 mL/kg, but some patients may need as much as 350 mL/kg.
Continue to reassess the patient for signs of dehydration at least every 4 hours to ensure that enough ORS solution is being taken. Patients with profuse ongoing diarrhea require more frequent monitoring. If signs of some dehydration are detected, the patient should be rehydrated as described earlier, before continuing with treatment to maintain hydration.
A few patients, whose ongoing stool output is very large, may have difficulty in drinking the volume of ORS needed to maintain hydration. If these patients become tired, vomit frequently, or develop abdominal distension, ORS solution should be stopped and hydration should be maintained intravenously with lactated Ringer solution or isotonic sodium chloride solution, administering 50 mL/kg in 3 hours. After this is done, resuming treatment with ORS solution is usually possible.
Keep the patient under observation, if possible, until diarrhea stops or is infrequent and of small volume. This is especially important for any patient presenting with severe dehydration. If a patient must be discharged from the hospital before diarrhea has stopped, show the caretaker how to prepare and give ORS solution, and instruct the caretaker to continue to give ORS solution, as above. Also instruct the caretaker to return the patient to the hospital if any signs of danger appear.
Antibiotic Treatment in Cholera;
An effective antibiotic can reduce the volume of diarrhea in patients with severe cholera and shorten the period during which V cholerae O1 is excreted.
In addition, it usually stops the diarrhea within 48 hours, thus shortening the period of hospitalization. Whenever possible, antibiotic therapy should be guided by susceptibility reports.
Antibiotic treatment is indicated for severely dehydrated patients who are older than 2 years. Begin antibiotic therapy after the patient has been rehydrated (usually in 4-6 h) and vomiting has stopped. No advantage exists to using injectable antibiotics, which are expensive. No other drugs should be used in the treatment of cholera. Antimicrobial agents typically are administered for 3-5 days.
However, single-dose therapy with tetracycline, doxycycline, furazolidone, or ciprofloxacin has been shown effective in reducing the duration and volume of diarrhea. Because single dose doxycycline has been shown to be as effective as multiple doses of tetracycline, this has become the preferred regimen.
1. Doxycycline†
7 mg/kg; not to exceed 300 mg/dose
2 mg/kg bid on day 1; then 2 mg/kg qd on days 2 and 3; not to exceed 100 mg/dose
2. Tetracycline
25 mg/kg; not to exceed 1 g/dose‡
40 mg/kg/d divided qid for 3 d; not to exceed 2 g/d
3. Trimethoprim and sulfamethoxazole
< 2 months of age: CTX is Contraindicated
≥2 months: 5-10 mg/kg/d (based on trimethoprim component) divided bid for 3 d; not to exceed 320 mg/d trimethoprim and 1.6 g/d of sulfamethoxazole
4. Ciprofloxacin
30 mg/kg; not to exceed 1 g/dose
30 mg/kg/d divided q12h for 3 d; not to exceed 2 g/d
5. Ampicillin
50 mg/kg/d divided qid for 3 d; not to exceed 2 g/d
6. Erythromycin
40 mg/kg/d erythromycin base divided tid for 3 d; not to exceed 1 g/d.
Antimicrobial therapy is an adjunct to fluid therapy of cholera and is not an essential component. However, it reduces diarrhea volume and duration by approximately 50%. The choice of antibiotics is determined by the susceptibility patterns of the local strains of V cholerae O1 or O139.
Diet in Cholera;
Resume feeding with a normal diet when vomiting has stopped. Continue breastfeeding infants and young children.
Malnutrition after infection is not a major problem, as it is after infection with Shigella species or rotavirus diarrhea. The catabolic cost of the infection is relatively low, anorexia is neither profound nor persistent, and intestinal enzyme activity remains intact after infection; hence, intestinal absorption of nutrients is near normal.
There is no reason to withhold food from cholera patients.
Prevention of Cholera;
1. Food and water sanitation.
2. Screening of food handlers.
3. In outbreaks a single dose of Doxycycline 300mgs provides adequate prophylaxis to those exposed or at risk with no clinical disease.
Click here to access Unit Two Content..
Topic 1.4: DYSENTERY
The term dysentery refers to any bloody stool or blood in stool.
There are two common types of dysentery;
1. Amoebic dysentery- Mostly caused by Entamoeba Histolytica.
2. Bacilliary dysentery- Also known as Shigellosis, caused by Shigella Dysenteriae.
Background
Shigellosis occurs when Shigella species invade the epithelial lining of the terminal ileum, colon, and rectum, causing diarrhea and bacillary dysentery that ranges from mild to severe disease. It is a major public health problem in developing countries where sanitation is poor but also exists in industrialized nations among the homeless and those with low and middle income.
Aetiology;
Shigella organisms cause bacillary dysentery, a disease that has been described since early recorded history.
Shigella species dysenteriae,
1. S flexneri,
2. S sonnei,
3. S boydii
Are aerobic, nonmotile, glucose-fermenting, gram-negative rods that are highly contagious, causing diarrhea after ingestion of as few as 180 organisms.
Shigellosis is highly infectious and spreads by fecal-oral transmission. Shigella sonnei and Shigella flexneri cause 90% of the cases of shigellosis; Shigella dysenteriae has produced epidemic shigellosis. Left untreated, fulminant dysentery may occur and is potentially fatal. No vaccine against Shigella species exists yet, however, several are under development.
Epidemiology;
Shigellosis occurs worldwide, and it tends to occur whenever war, natural calamities (eg, earthquakes, floods), or unhygienic living conditions result in overcrowding and poor sanitation.
S boydii and S dysenteriae occur more commonly internationally. Disease from Shigella species causes an estimated 700,000 deaths and 165 million cases of diarrhea annually worldwide.
Mode of transmission;
Shigellosis is spread by means of fecal-oral transmission.
The organism is spread by fecal-oral contact via infected food or water, during travel or in long-term care facilities, daycare centers, or nursing homes.
Risk factors;
1. Children in daycare,
2. Incarcerated persons,
3. International travelers,
4. Homosexual men,
5. People infected with human immunodeficiency virus (HIV)
6. Crowded, unsanitary conditions.
Shigellosis follows a self-limited course ranging from 3 days to 1 week. It rarely lasts as long as a month.
Shigellosis is most common in children aged 6 months to 5 years.
Pathophysiology;
Shigella infection occurs essentially through oral contamination via direct fecal-oral transmission, the organism being poorly adapted to survive in the environment. Resistance to low-pH conditions allows shigellae to survive passage through the gastric barrier, an ability that may explain in part why a small inoculum (as few as 100 CFU) is sufficient to cause infection.
The organisms induce apoptosis of subepithelial resident macrophages. Once inside the cytoplasm of intestinal epithelial cells, Shigella effectors trigger the cytoskeletal rearrangements necessary to direct uptake of the organism into the epithelial cell. The Shigella-containing vacuole is then quickly lysed, releasing bacteria into the cytosol.
Intracellular shigellae next use cytoskeletal components to propel themselves inside the infected cell; when the moving organism and the host cell membrane come into contact, cellular protrusions form and are engulfed by neighboring cells. This series of events permits bacterial cell-to-cell spread.
Cytokines released by a growing number of infected intestinal epithelial cells attract increased numbers of immune cells [particularly polymorphonuclear leukocytes (PMNs)] to the infected site, thus further destabilizing the epithelial barrier, exacerbating inflammation, and leading to the acute colitis that characterizes shigellosis. Evidence indicates that some type III secretion system–injected effectors can control the extent of inflammation, thus facilitating bacterial survival.
Shiga toxins are translocated from the bowel into the circulation. After binding of the toxins to target cells in the kidney, pathophysiologic alterations may result in hemolytic-uremic syndrome.
Signs and Symptoms of shigellosis :
Manifestations of shigellosis generally begin within 1-2 days of infection and can include a range of the following signs and symptoms :
1. Acute bloody diarrhea
2. Crampy abdominal pain
3. Tenesmus
4. Passage of mucus
5. Fever (1-3 days after exposure)
6. Occasionally vomiting (35% prevalence)
7. Self-limited course (3 days to 1 week and, rarely, lasts as long as 1 month)
Physical examination;
1. Lower abdominal tenderness
2. Normal or increased bowel sounds
3. Dehydration (occasional)
Diagnosis;
1. Stool analysis;
The "gold standard" for the diagnosis of Shigella infection remains the isolation and identification of the pathogen from fecal material.
One major difficulty, particularly in endemic areas where laboratory facilities are not immediately available, is the fragility of Shigella and its common disappearance during transport, especially with rapid changes in temperature and pH. In the absence of a reliable enrichment medium, buffered glycerol saline or Cary-Blair medium can be used as a holding medium, but prompt inoculation onto isolation medium is essential.
The probability of isolation is higher if the portion of stools that contains bloody and/or mucopurulent material is directly sampled. Rectal swabs can be used, as they offer the highest rate of successful isolation during the acute phase of disease.
A sample of stool for culture should be obtained in all suspected cases of shigellosis. Specimens should be processed immediately after collection.
2. Full haemogram; may be performed in persons with severe symptoms or to rule out other causes.
Diagnostic Procedure;
Sigmoidoscopy is not necessary in most cases of shigellosis.
If distinguishing between dysentery and the acute presentation of idiopathic ulcerative colitis is urgently necessary, a colonic biopsy may be useful if it is performed within 4 days of the onset of symptoms. Histologic findings of shigellosis include the following:
1. Intense neutrophilic and mononuclear cells infiltrating the lamina propria
2. Hemorrhage
3. Ulcers
4. Mucous depletion
5. Occasional crypt abscesses
Complications of Shigellosis;
Complications from shigellosis may be local/intestinal or systemic.
1) Intestinal complications;
i. Proctitis or rectal prolapse is common in infants and young children. This is induced by invasion of the organism into the colonic mucosa, causing severe inflammation of the rectum and distal colon.
ii. Toxic megacolon occurs primarily in the setting of S dysenteriae infection. The pathogenesis is unclear, but it occurs in patients with pancolitis and seems to be related to the intensity of inflammation rather than being mediated by the Shiga toxin.
iii. Intestinal obstruction can develop from severe colonic disease. The patients with obstruction are more likely to be infected with type 1 S dysenteriae.
iv. Colonic perforation is an extremely rare complication of shigellosis. It occurs primarily in infants or severely malnourished patients and is associated with infection due to type 1 S dysenteriae or S flexneri.
2) Systemic complications;
i. Shigella bacteremia has a reported incidence of 0-7%. Signs that correlate with bacteremia are;
a) leukocytosis,
b) hypothermia,
c) Fever; temperature above 39.5ºC,
d) severe dehydration, and
e) lethargy.
Bacteremia is more common among children than in adults, occurring primarily in those younger than 5 years. In one study, among the 22 cases of bacteremia described in adults, one third of patients were older than 65 years, and more than half had an underlying disease (most commonly diabetes). However, infection with human immunodeficiency virus (HIV) does not appear to confer a significant predisposition to Shigella bacteremia.
Differential Diagnoses
1. Amebiasis
2. Bacterial Gastroenteritis
3. Campylobacter Infections
4. Cholera
5. Clostridioides (Clostridium) Difficile Colitis
6. Colon Cancer
7. Crohn Disease
8. Cryptosporidiosis
9. Escherichia coli (E coli) Infections
10. Pseudomembranous Colitis
11. Salmonella Infection (Salmonellosis)
12. Ulcerative Colitis
13. Viral Gastroenteritis
14. Yersinia Enterocolitica.
Management of Shigellosis;
General supportive care of patients with shigellosis includes the following:
1. Pyrexia management
High fever in children should be treated.
2. Clear liquids followed by a low-residue, lactose-free diet are recommended until symptoms of shigellosis resolve.
3. For fluid and electrolyte supplementation, oral rehydration solutions are preferable.
4. Educate patients about proper hygiene. Careful handwashing and stool precautions should prevent the dissemination of shigellosis. Thus, primary preventive measures should include universal availability of potable water, provision of sanitation methods, and improved personal and food hygiene
Drugs to avoid in shigellosis;
i. Narcotic-related antidiarrheals should be avoided due to risk of paralytic ileus and toxic megacolon.
ii. Antimotility agents should be avoided. They have the potential to worsen symptoms and may predispose to toxic dilation of the colon.
Antibiotic treatment;
This is indicated in most patients. Note that antibiotic-resistant Shigella species have emerged; thus, obtaining susceptibility testing and monitoring local shigellosis outbreaks is crucial.
Given the widespread resistance to ciprofloxacin as well as trimethoprim-sulfamethoxazole and azithromycin, a third-generation cephalosporin is appropriate empiric therapy in the setting of acute illness. The treatment of choice for HIV-infected patients is a quinolone for 5 days.
1. Ciprofloxacin.
2. Ceftriaxone.
3. Azithromycin.
Prevention of Shigellosis;
A vaccine for shigellosis is not currently available. Until a vaccine is available, the following measures can help prevent the dissemination of shigellosis:
1) Use of safe drinking water
2) Chlorination of unreliable water sources
3) Strict handwashing
4) Refrigeration and proper preparation and cooking of food. Food handlers must be treated with antibiotics and should not be involved in food preparation as long as stool cultures are positive for Shigella infection. At least 48 hours of antibiotic treatment are usually required.
Click here to access Unit Two Content..
Topic 1.5: TYPHOID FEVER
Click here to access Unit Two Content..
Topic 1.6: PLAGUE/YERSINNIOSIS
The genus Yersinia currently contains 11 species, only 3 of which are known to be significant human pathogens
1. Y. pestis,
2. Y. enterocolitica, and
3. Y. pseudotuberculosis
Although these minor yersiniae have been identified primarily from environmental samples, they have occasionally been recovered from patient isolates, thus suggesting possible roles as human pathogens.
Y. PESTIS /PLAGUE;
Plague is a highly fatal, flea-borne disease that is best known as the cause of the Black Death of the Middle Ages.
Its etiologic agent, Y. pestis, is a gram-negative coccobacillus belonging to the family Enterobacteriaceae.
Epidemiology;
Y. pestis is maintained in nature through transmission cycles involving certain rodent species and their fleas, which act as vectors.
Although other mammals often become infected with Y. pestis and occasionally succumb to plague, the only nonrodent species thought to be important as hosts for infecting vector fleas are some species of rabbits and hares, the steppe pika of central Asia, and the house shrew of southeastern Asia and Madagascar.
Rodent-consuming carnivores and raptors might also play indirect roles in spreading plague by transporting infected rodent fleas from one area to another.
Humans most frequently acquire plague as a result of being bitten by infectious fleas. On a worldwide basis, the risk of flea bite exposure is highest in poverty-stricken areas that are situated near natural plague foci and have large infestations of commensal rats (Rattus sp) heavily infested with fleas, particularly the oriental rat flea (Xenopsylla cheopis). X. cheopis readily feeds on humans and is an efficient vector of Y. pestis to people, as well as rats.
Persons are most likely to be bitten by infectious X. cheopis when plague epizootics cause massive mortality among susceptible rats, thereby forcing these fleas to seek new hosts. Currently, certain regions of central and southern Africa (including Madagascar), southeastern Asia and India, and a few areas in South America remain at high risk for rat-associated plague outbreaks.
The spread of rat-associated plague from one region to another, perhaps by natural movement of rats or transport of these animals along with trade goods, poses the threat that major epidemics will occur in large rat-infested cities. The appearance of bubonic cases acquired via flea bite in large cities also greatly increases the threat that pneumonic plague will develop in untreated bubonic cases and that the infection will spread to others through coughing and expulsion of infectious respiratory droplets (primary pneumonic plague).
Recently, concern has been raised that plague could be used as an agent of bioterrorism. In most projected scenarios, bioterrorists would spread plague in an aerosol form, potentially resulting in numerous primary pneumonic cases, high mortality, and widespread panic, especially if the Y. pestis strains released have been engineered to be resistant to antimicrobial agents commonly used to treat plague.
Few bacteria can rival the pathogenicity of Y. pestis for humans. Y. pestis usually enters the body at the site of a flea bite or perhaps as a result of contact between abraded or cut skin and blood or other body fluids from a Y. pestis–infected animal.
Immunobiology of yersiniosis;
On entering the body, plague bacteria come under attack by host phagocytes and other host defenses. The ability to escape from these defenses and disseminate to regional lymph nodes depends in part on a protease (Pla) that helps degrade fibrin clots and promote the production of excess plasmin that can affect inflammatory exudates, break down extracellular proteins and basement membranes, and reduce levels of chemoattractants, possibly because of inhibition of interleukin-8 production at the site of initial infection.
Affected phagocytes are rendered incapable of killing the invading Y. pestis, thereby allowing this bacterium to survive extracellularly in lymphoid tissues. Survival of Y. pestis in mammalian hosts also depends on its ability to acquire sufficient quantities of iron for growth.
The most important means of iron uptake in Y. pestis is a siderophore (yersiniabactin) system that can effectively compete with host iron-binding molecules for this essential nutrient. Although the ability of Y. pestis to resist host phagocytic killing and survive intracellularly in phagocytes is thought to be particularly important during the early stages of infection, plague bacteria also express a glycoprotein capsular antigen (caf1 or fraction 1 antigen) that confers resistance to phagocytosis. Expression of caf1 is temperature dependent, being repressed at the cooler temperatures found in the flea vector and upregulated at mammalian host body temperatures.
Buboes;
Infected lymph nodes, termed buboes, can appear edematous and congested early in the course of illness but exhibit little evidence of inflammatory infiltrates or vascular injury. These buboes represent the most obvious manifestation of the lymphatic system's efforts to arrest the spread of Y. pestis, and within a few days after infection they contain massive numbers of Y. pestis and heavy neutrophil infiltrates, which causes them to swell to the size of a hen's egg and become surrounded by serous fluid.
As illness progresses, hemorrhagic necrosis and vascular damage in the node become apparent; some nodes spontaneously rupture, and abscesses appear. Although Y. pestis can be present in small quantities in blood samples taken relatively early in the course of infection, large quantities of plague bacteria usually appear in the blood of patients with bubonic plague only after the lymph node defenses are overwhelmed.
As the bacteria escape from the node and proliferate in blood, patients with bubonic plague begin to exhibit evidence of plague septicemia (secondary septicemic plague). Patients with inadequately treated septicemic plague can experience widespread and overwhelming destruction of tissues as Y. pestis spreads to various organs, eventually resulting in their failure.
Cause of death in plague;
Patients who die of plague often experience;
1. Diffuse interstitial myocarditis,
2. Cardiac dilation,
3. Diffuse hemorrhagic splenic necrosis,
4. Renal glomeruli containing fibrin thrombi,
5. Multifocal necrosis in the liver.
6. Disseminated intravascular coagulation-DIC
DIC can lead to thrombosis within capillaries, vascular necrosis, ecchymoses, acral gangrene, and cutaneous, mucosal, and serosal petechiae. In a small proportion of cases, septicemia occurs in the absence of buboes or other signs of localized infection, a condition referred to as primary septicemic plague. Septicemic plague can also occur secondarily to primary pneumonic plague.
Rarely, plague can be acquired through the inhalation of infectious respiratory droplets or other materials. These patients with primary pneumonic plague typically experience a rapidly progressing lung infection that is at first lobular, then lobar, and finally multilobar, with large numbers of Y. pestis being present in the alveoli and pulmonary secretions of affected sites. Pneumonic plague can also occur secondarily to bubonic or primary septicemic plague.
Clinical features of plague;
The three most commonly observed forms of plague (listed in order of decreasing occurrence) are bubonic, septicemic, and pneumonic. Unusual manifestations of plague include meningitis and pharyngitis. In rare instances, Y. pestis has also been inoculated through the conjunctiva, thereby resulting in oculoglandular plague. The incubation period is 2 to 6 days for bubonic plague and 1 to 3 days for primary pneumonic plague.
1. Bubonic plague;
i. Swollen and tender lymph nodes (buboes) of bubonic plague usually appear in the node or nodes located closest to the site of initial infection as indicated by the appearance of inguinal or femoral lymph node involvement on the side where the flea bite initially occurred.
Axillary buboes also commonly occur, often indicating handling of an infected animal or other flea-infested object.
Symptoms of bubonic plague include
1) Fever,
2) Chills,
3) Myalgia,
4) Arthralgia,
5) Headache,
6) Malaise, and
7) Prostration.
Untreated patients with bubonic plague become increasingly toxic, remain febrile, and experience tachycardia, agitation, confusion, delirium, and convulsions.
2. Septicemic plague is manifested as;
1) Rapidly progressive, overwhelming endotoxemia.
2) Gastrointestinal symptoms, including
i. Nausea,
ii. Vomiting,
iii. Diarrhea, and
iv. Abdominal plain.
3) Disseminated intravascular coagulation can also occur with the appearance of petechiae, ecchymoses, bleeding, and ischemia in the tips of the extremities.
4) Later-stage septicemic patients are likely to experience refractory hypotension, renal shutdown, obtundation, and other signs of shock.
5) Patients with late-stage septicemic plague can also exhibit adult respiratory distress syndrome, which has occasionally been confused with hantaviral pulmonary syndrom.
Because septicemic plague is likely to be fulminant and fatal, favorable outcomes depend on rapid diagnosis and prompt treatment with appropriate antimicrobials.
3. Pneumonic plague can be accompanied by;
i. Fever,
ii. Cough,
iii. Chest discomfort that becomes increasingly painful,
iv. Tachycardia,
v. Dyspnea,
vi. Bacteria-laden sputum, chills, fever, headache, achiness, weakness, and dizziness.
As the illness progresses, patients can also experience increasing respiratory distress, hemoptysis, cardiopulmonary insufficiency, and circulatory collapse. Patients with primary pneumonic plague in the early stages of illness can have signs of localized pulmonary involvement, beginning in a single lung and rapidly progressing to segmental consolidation and later bronchopneumonia, with death ensuing within 24 hours of the onset of symptoms.
Localized infection is unlikely to be observed in the lungs of patients with secondary pneumonic plague because the lung tissues are infected initially through circulatory spread, which results in a diffuse interstitial pneumonitis. The appearance of sputum also differs between primary and secondary pneumonic plague, being watery or mucoid, frothy, and perhaps tinged with blood in primary pneumonic cases but scanter, thicker, and more tenacious in secondary pneumonic cases.
Laboratory diagnosis;
1. Bacterial culture
2. Specific bacteriophage lysis tests
3. Detection of a four-fold rise in antibody titer to the Y. pestis F1 capsular antigen over a period of 2 to 4 weeks.
Direct fluorescent antibody assays can be used to identify Y. pestis bacteria in bubo aspirates, sputum samples, and tracheal washes; this procedure requires about 1 hour and provides strong presumptive evidence of infection.
A presumptive diagnosis can also be obtained rapidly by detecting Y. pestis DNA in polymerase chain reaction (PCR) assays or Y. pestis–specific antigens in immunologic assays, but these tests are not widely available.
Preferred laboratory samples include blood, serum, bubo aspirates, tracheal washes, and swabs of skin lesions or pharyngeal mucosa. Cerebrospinal fluid can also be collected from patients in whom plague meningitis is suspected.
Treatment of Plague;
i. The most commonly recommended agent for treating plague is streptomycin.
ii. A recently completed randomized trial in Tanzania concluded that gentamicin and doxycycline were effective for treating plague in adults and children and caused few adverse responses.
A recent review of human cases treated in New Mexico also strongly suggested that gentamicin is effective and can be substituted for streptomycin. Tetracyclines are effective for treating uncomplicated cases of bubonic plague, and chloramphenicol is believed to be effective, particularly for plague meningitis.
The occasional persistence of Y. pestis in necrotic or partially necrotic tissues emphasizes the importance of adequately excising these tissues.
Other drugs include;
iii. Chloramphenicol.
iv. Ciprofloxacin.
New Programs
- International Studies
- Models & Language Reaching
- Public School Facts
- State Testing Data
- Education Jobs
Latest News
OCTOBER, 2021
We start our academic term, with a very determine pace...
December, 2021
We release our results...


